 |
 |
| Anim Biosci > Volume 30(2); 2017 > Article |
|
Abstract
Objective
The study was conducted to evaluate the stability of commercial coated lipase (CT-LIP) in vitro.
Methods
The capsules were tested under different conditions with a range of temperature, pH, dry heat treatment and steaming treatment, simulated gastric fluid (SGF) and simulated intestinal fluid (SIF) in this work, respectively. Free lipase (uncoated lipase, UC-LIP) was the control group. Lipase relative activities measured in various treatments were used as a reference frame to characterize the stability.
Results
The lipase activities were decreased with increasing temperatures (p<0.05), and there was a markedly decline (p<0.01) in lipase comparative activities of UC-LIP at 80°C compared with CT-LIP group. Higher relative activities of lipase were observed in CT-LIP group compared with the free one under acidic ambient (pH 3 to 7) and an alkaline medium (pH 8 to 12). Residual lipase activities of CT-LIP group were increased (p<0.05) by 5.67% and 35.60% in dry heat and hydrothermal treatments, respectively. The lipase relative activity profile of CT-LIP was raised at first and dropped subsequently (p<0.05) compared with constantly reduced tendency of UC-LIP exposed to both SGF and SIF.
The stress of weaning and immature digestive tract makes young pigs susceptible to considerable challenges, such as digestive disorders, diarrhea, higher morbidity and mortality [1,2]. Nutritive antibiotics could improve growth rate and efficiency of feed utilization, reduce morbidity and mortality, and improve reproductive performance [3]. However, antibiotic-resistant pathogens are linked with the use of antibiotics as growth promoters which limits their utility in livestock husbandry [2]. Exogenous enzymes proposed as alternatives to antibiotics, which include carbohydrase, lipase, protease, and phytase, are now extensively used throughout the world as feed additives in monogastric animal diets [4–8].
Lipases are renowned for their versatility in addition to their ability to digest fat. They can be employed for the formulation of detergents, as feed ingredients and as biocatalysts [9]. Jensen et al [10] investigated the development of lipases in pigs and noted a pronounced decrease in pancreatic and an increase in stomach lipase activity (only 0.2% of the pancreatic activity) in newly weaned piglets. Low activity of pancreatic lipase and intestinal lipase in piglets during the first week after birth or post weaning may be one important cause of low-fat digestibility in neonatal or weaning piglets [11]. Some studies have shown that the combined use of triacylglycerols containing medium-chain fatty acids or fatty powder and exogenous lipolytic enzymes could improve growth performance [12], carcass composition [13], antimicrobial activity, clearly regulate and stabilize the gastrointestinal flora in the stomach [14].
However, the crucial disadvantages of feed in stability, odour and storage caused by the endogenous lipase preparations limits their utility as feed additives in livestock production [15]. Microcapsules are promising candidates for encapsulating, delivering, and controllably releasing many technologically important actives, including agricultural chemicals, food additives, and pharmaceuticals [16]. The objective of our study was to assess the stability of the commercial encapsulated lipase (coated lipase, CT-LIP) in vitro.
The following materials and chemicals were obtained from commercial suppliers: CT-LIP (lipase from Bacillus stearothermophilus, 9,012 U/g, Zhuhai Tiankai Biochemical Co. Ltd., Guangdong, China), uncoated lipase (UC-LIP, from Bacillus stearothermophilus, 10,935U/g, Zhuhai Tiankai Biochemical Co. Ltd., Guangdong, China), pepsin (from porcine, Sigma P7012, St. Louis, MO, USA), pancreatin (from porcine, Sigma P1750, USA). The enzyme activity unit (1 U) is defined as the amount of the lipase that catalyzes the production of one micromole (1 μmol) free fatty acid per minute under certain conditions. All other chemicals and solvents used were of analytical grade.
The CT-LIP and UC-LIP were subjected to a diverse series conditions such as different pH and temperatures, dry heat treatment and hydrothermal process, and simulated gastrointestinal (SGI) tract in this work. The UC-LIP was invoked as the control group. Lipase relative activities detected in various treatments were used as a reference frame to characterize the stability of enzymes. The lipase relative activity profile of CT-LIP and UC-LIP samples in multiple conditions was reported. Each treatment was performed in six replications.
The emulsion used for assaying enzyme activity was prepared freshly. In brief, the solution (1,000 mL) with 40 g/L polyvinyl alcohol (polymerization degree 1,750±50, Guangcheng chemical Co., Tianjin, China) was filtered through double-layer gauze, and then the olive oil (50 mL) was mixed with filter liquor (1/3, v/v). The resulting liquid was homogenized immediately for 6 min with high-speed homogenizer to ensure uniform dispersal.
The lipase activities of CT-LIP and UC-LIP were determined at 37°C in this study, using the titrimetric method as previously reported [17]. Briefly, the emulsion (4.00 mL) and phosphate buffer saline (PBS, pH 7.5, 5.00 mL) were added to blank conical flask (marked A) and trialed conical flask (marked B), respectively. In addition, another 15.00 mL 95% (v/v) ethanol solution was added to the A conical flask and heated in a water bath (T 42°C±0.2°C) for 5 min. Subsequently, equal enzyme solution treated by different conditions was added to A and B system, separately. Reaction in B system was terminated by adding 15.00 mL 95% (v/v) ethanol solution after 15 min reaction time. The free fatty acids were titrated against standard NaOH solution indicated with phenolphthalein. The lipase activity and its relative activity were determined by following equations.
Where Xd is detected enzyme activity (u/g) in each sample, V1 (mL) and V2 (mL) were the volume of NaOH solution consumed by B and A trail, respectively. The character c (M) and n indicates the concentration of NaOH solution in titration and the dilution ratio of samples. Reaction time (character t) is 15 min in this experiment. Rx and Xm express the lipase relative activity and the maximum lipase activity detected in the same condition, respectively. The measured value in each time point was compared with the highest activity in control and experimental reactions to obtain the relative activity. The CT-LIP properties such as thermal stability, pH stability, tolerance of heat treatment and SGI tract environment were characterized as follows.
Samples (CT-LIP and UC-LIP, 1.0000 g) diluted with freshly prepared PBS (pH 7.5, 1,000 mL) were incubated at 40°C, 50°C, 60°C, 70°C, 80°C, 90°C, and 100°C for 30 min. After incubating, the solution was cooled down in an ice water bath and the pH was adjusted to 7.5. Formed resulting mixtures were immediately homogenized for 3 min with high-speed homogenizer, and homogenate was used to assay enzyme activity.
CT-LIP and UC-LIP (1.0000 g) were placed in 1,000 mL different PBS with pH range of 3.0 to 12.0 and then incubated at 40°C water bath for 30 min. The buffer solutions were prepared with the following formulations. Formed resulting mixtures were immediately homogenized for 3 min with high-speed homogenizer, and homogenate was used to assay enzyme activity. Different formulations prepared in this study were shown in Table 1.
Effect of dry heat treatment on CT-LIP was conducted at 80°C for 0.5 h by using electric constant temperature drying oven, while hydrothermal process of CT-LIP was performed in portable high-pressure steam sterilizer with 100°C saturated vapor for 4 min. Samples were cooled to room temperature after multiple treatments. The relative lipase activity was analyzed before and after experiments.
The activity profile of CT-LIP in SGI tract containing enzymes was also evaluated, using the methods as previously described [18–20]. Briefly, treatment of samples (CT-LIP or UC-LIP) in SGI tract conditions refers to dissolution carried out in simulated gastric fluid (SGF, pH 3.5) and simulated intestinal fluid (SIF, pH 6.8) at 39°C (water bath), respectively. Samples (CT-LIP or UC-LIP, 0.5 g) were incubated in 400 mL SGF with 1% pepsin (w/v) separately. The mixtures were incubated at 39°C in a shaking water bath for 1, 2, 3, 4, and 5 h. Similarly, the samples (CT-LIP or UC-LIP, 0.5 g) were digested by 400 mL of SIF with 1% pancreatin (w/v) at 39°C under mild stirring for 1, 2, 3, 4, 5, and 6 h, respectively. The digestion above was stopped by raising the pH to 7.5 with NaOH or HCl. The activity was investigated as aforementioned.
Statistical analyses of the data were performed using SPSS 17.0 software (SPSS Inc., Chicago, IL, USA). Statistical differences between means were tested using one way analysis of variance (ANOVA) or t-test, where appropriate. For each experimental series, statistical differences were considered significant with p<0.05 whereas 0.05≤p<0.10 was considered a tendency. The t-test was used to analyze dry heat and hydrothermal stability of CT-LIP and to determine the stability difference between CT-LIP and UC-LIP under the same conditions. The statistical model one-way ANOVA was used to analyze the thermal stability, pH stability, and tolerance in SGI tract:
Where Yij is the measured variable, μ is an overall mean, αi - the different conditions effect (treatment), and eij - the random error.
As shown in Figure 1, the lipase activities were decreased with increasing temperatures (p<0.05), and there was a markedly decline in lipase relative activities of UC-LIP compared with CT-LIP group (p<0.01). At 80°C, an extremely significant difference (p<0.01) was observed between the activities of the CT-LIP (65.80%) and UC-LIP (32.30%) group. High temperature causes inactivation of conventional enzymes. As the temperature rose, CT-LIP protected by coating showed a stronger thermostability than the free one. The stability is a key factor in estimating the economic feasibility of commercial enzymes [21,22]. High pressure steam, which provides the appropriate heat and moisture required for thermal modification of starch and protein in pellet processing, would produce certain damage to the activity of supplemented lipase. Conditioning the feed at a temperature of 80°C, in which CT-LIP has more heat stability, is sufficient to produce good quality pellets. Therefore, upon keeping its action during pellet processing, CT-LIP could have a considerable effect on the feed industry. Superior stability of enzyme preparations in pellets can withstand repeated handlings in the feed industry, such as bagging, transportation, and storage. CT-LIP encapsulation as the physical enclosure of the enzyme within a polymeric membrane [23] could reduce the degradation of fat in feedstuff catalyzed by additive lipase and therefore improve the storage capacity.
The pH is one of the important parameters of enzyme stability in aqueous solution. As shown in Figure 2, more of the higher relative activities of lipase were observed in CT-LIP group compared with the free one under acidic ambient (pH 3 to 7) and an alkaline medium (pH 8 to 12). There is a similar trend in the relative activity of the two enzyme preparations. Under various conditions of pH range from 3 to 12, the both activities increased first (p<0.05), dropped subsequently (p<0.05) and reached the highest value at pH 8.0. The results observed from this work are consistent with the report that the lipase purified from the culture supernatant of a thermophilic Bacillus sp. is optimally active at 60°C and pH 8.0 [24].
Similar results have been reported by Kenthorai et al [25], which also suggested that the enzyme activity increased first and then declined under different pH conditions. The difference was that Kenthorai et al [25] has shown that the enzyme activities of the encapsulated were always higher than the free. This may be attributed to the different source of lipase or the encapsulating materials. Generally, the CT-LIP had a stronger acid resistance than UC-LIP. The CT-LIP may retain the potential property to provide sustained release of lipase or resistance to acidic environment and thus improved its bioavailability in the upper part of small intestine.
As shown in Figure 3, residual lipase activities of CT-LIP group were increased by 5.67% (p<0.05) and 35.60% (p<0.05) in the dry heat and hydrothermal treatments, respectively, compared with UC-LIP group. Residual enzyme activity of CT-LIP was 80.26% through the dry heat process and was 59.97% suffered hydrothermal treatment. Greater dry and damp heat stability was observed significantly in CT-LIP treatments compared with the control group (p<0.05).
The heat tolerance of the CT-LIP was enhanced according to our research. Hydrothermal and dry heat treatments could damage the three-dimensional structures the of enzyme preparations during pelleted process, and therefore cause inactivation of enzymes. Inactivation of enzymes would occur partially at pelleting, and the time of conditioning and higher temperature process would expedite inactivation [26]. In this work, the encapsulation materials provided protective actions to the enzyme structure.
The lipase relative activity profile of CT-LIP was increased at first and dropped subsequently (p<0.05) compared with constantly reduced tendency of UC-LIP exposed to both SGF (p = 0.078) and SIF (p = 0.069) in the present study.
As shown in Figure 4a, the relativity activity profile rose from 61.93% to 100% through the first 4 h in CT-LIP group. The relative activity of UC-LIP declined constantly from 53.41% to 35.79%. As shown in Figure 4b, during experimental times, the relative activity of CT-LIP reached the higher peak at 2 h and then exhibited a general decline tendency. However, the relative activity of UC-LIP declined significantly from 94.98% to 65.73% in the first two hours and then appeared slow reduction.
The treatment of SGI tract was performed in order to assess the protective effect of coating against gastrointestinal conditions in vitro. When exposed to an acidic pH<7.0 (SGF), the microcapsule of CT-LIP caused the coating shells to dissolve at a constant rate, starting at their exteriors. After a time delay of four hours, the shells became fully dissolved and the microcapsules released lipase. These results indicated that CT-LIP had a stronger resistance than UC-LIP in SGF. Su et al [27] reported that degradation of coating phytase by pepsin was obviously lessened. The additional trypsin or proteolytic enzyme could provoke a significant reduction in the activity of free lipase and little influence on the CT-LIP [28]. When exposed to SIF, the relative activity of CT-LIP was increased (p<0.05) at 2 h, and then the relative activities of UC-LIP had a constant decline that was not significant. The difference between SGF and SIF may be attributed to digestive enzymes and pH in the gastrointestinal tract. We expect CT-LIP to take longer to fully dissolve and ultimately release lipase in the intestine of piglets.
The CT-LIP possesses relatively higher stability in comparison with the UC-LIP in vitro. The CT-LIP could potentially provide sustained release of lipase and thus improve its bioavailability in the gastrointestinal tract. A theoretical understanding of these properties would be very useful in the livestock industry.
ACKNOWLEDGMENTS
This article was financially supported by National “Twelfth Five-Year” Program for Science & Technology Support (2012BAD39B01).
Figure 1
Effect of different temperatures on lipase relative activity. Data were presented with mean±standard deviation (n = 6). Vertical error bars show standard deviation of measured relative activity (** p<0.01).
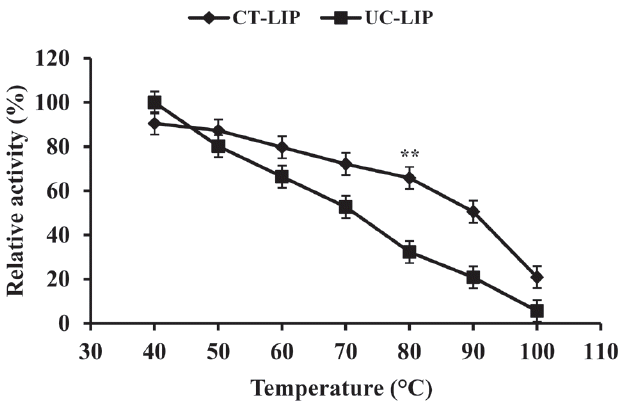
Figure 2
Effect of different pH on lipase relative activity. Data were presented with mean±standard deviation (n = 6). Vertical error bars show standard deviation of measured relative activity (* p<0.05).
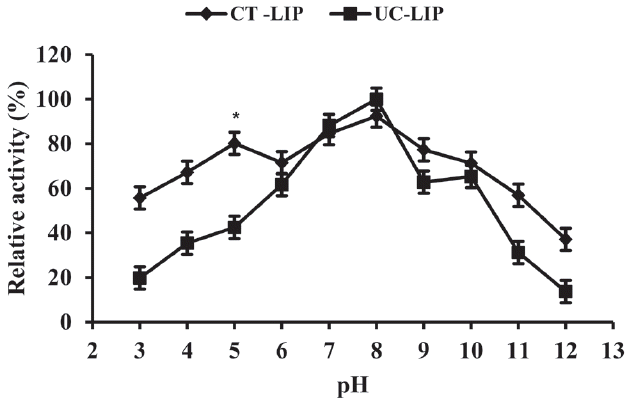
Figure 3
Comparative effects of dry heat treatment and hydrothermal process on the lipase relative residual activity of coated lipase (CT-LIP) and uncoated lipase (UC-LIP). Data were presented with mean±standard deviation (n = 6). Vertical error bars show standard deviation of measured relative activity (* p<0.05).
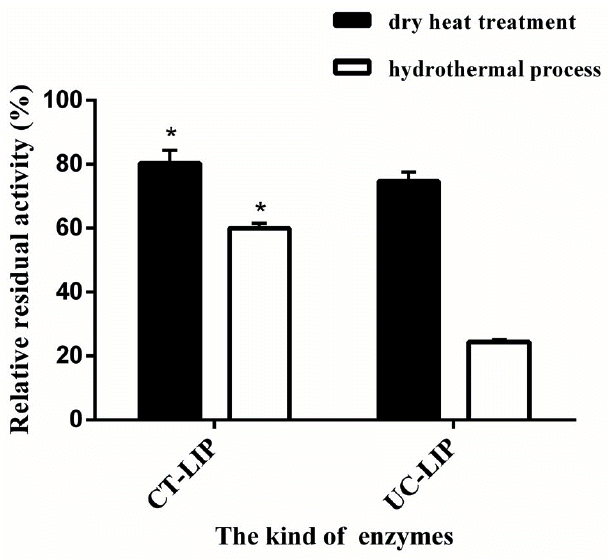
Figure 4
Effect of simulated gastrointestinal fluid on the lipase relative activity. (a) Effect of simulated gastric fluid (SGF) on the lipase relative activity. (b) Effect of simulated intestinal fluid (SIF) on the lipase relative activity. Data were presented with mean±standard deviation (n = 6). Vertical error bars show standard deviation of measured relative activity (* p<0.05).
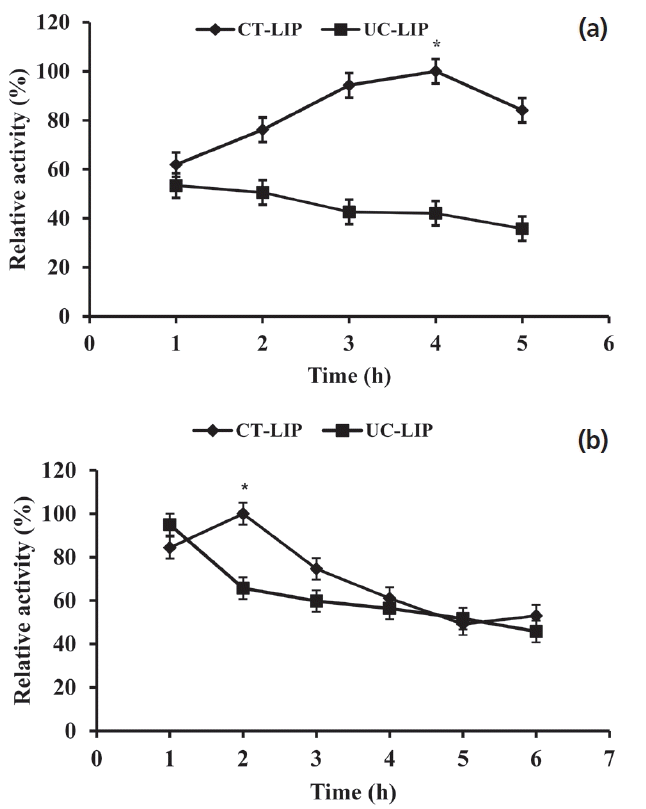
Table 1
Different formulations (100 mL) of complex solutions developed in this study
REFERENCES
1. Dierick NA, Decuypere JA, Molly K, Van Beek E, Vanderbeke E. The combined use of triacylglycerols (TAGs) containing medium chain fatty acids (MCFAs) and exogenous lipolytic enzymes as an alternative to nutritional antibiotics in piglet nutrition: II. In vivo release of MCFAs in gastric cannulated and slaughtered piglets by endogenous and exogenous lipases; effects on the luminal gut flora and growth performance. Livest Prod Sci 2002; 76:1–16.

2. Hong SM, Hwang JH, Kim IH. Effect of medium-chain triglyceride (MCT) on growth performance, nutrient digestibility, blood characteristics in weanling pigs. Asian-Australas J Anim Sci 2012; 25:1003–8.



3. Cromwell GL. Why and how antibiotics are used in swine production. Anim Biotechnol 2002; 13:7–27.


4. Roth RZ, Kirchgessner M. Organic acids as feed additives for young pigs: Nutritional and gastrointestinal effects. J Anim Feed Sci 1998; 7:25–33.

5. Partanen K, Mroz Z. Organic acids for performance enhancement in pig diets. Nutr Res Rev 1999; 12:117–45.


6. Bedford MR. Exogenous enzymes in monogastric nutrition-their current value and future benefits. Anim Feed Sci Technol 2000; 86:1–13.

7. Yin YL, McEvoy JDG, Schulze H, et al. Apparent digestibility (ileal and overall) of nutrients and endogenous nitrogen losses in growing pigs fed wheat (var. Soissons) or its by-products without or with xylanase supplementation. Livest Prod Sci 2000; 62:119–32.

8. Kiarie E, Romero LF, Nyachoti CM. The role of added feed enzymes in promoting gut health in swine and poultry. Nutr Res Rev 2013; 26:71–88.


9. Karjiban RA, Lim WZ, Basri M, Rahman MBA. Molecular dynamics of thermoenzymes at high temperature and pressure: a review. Prot J 2014; 33:369–76.

10. Jensen M, Jensen SK, Jakobsen K. Development of digestive enzymes in pigs with emphasis on lipolytic activity in the stomach and pancreas. J Anim Sci 1997; 75:437–45.


11. Gu X, Li D. Fat nutrition and metabolism in piglets: a review. Anim Feed Sci Technol 2003; 109:151–70.

12. Shi BL, Wang JY, Fu WY, Wang CY. Effect on Growth Performance of weaning piglets by microbe lipase. Feed Review 2010; 3:1–3.
13. Takada R, Shidara O, Saitoh M, Mori T. Effects of feeding medium chain triglycerides on growth performance, digestibility, backfat thickness and fatty acid composition in finishing pigs. Jpn J Swine Sci 1992; 29:32–40.

14. Decuypere JA, Dierick NA. The combined use of triacylglycerols containing medium-chain fatty acids and exogenous lipolytic enzymes as an alternative to in-feed antibiotics in piglets: concept, possibilities and limitations. Nutr Res Rev 2003; 16:193–210.


15. Dierick NA, Decuypere JA. Endogenous lipolysis in feedstuffs and compound feeds for pigs: effects of storage time and conditions and lipase and/or emulsifier addition. Anim Feed Sci Technol 2002; 102:53–70.

16. Abbaspourrad A, Datta SS, Weitz DA. Controlling release from pH-responsive microcapsules. Langmuir 2013; 29:12697–702.


17. Valivety R, Halling PJ, Peilow AD, Macrae AR. Lipase from different sources vary widely in dependence of catalytic activity on water activity. Biochim Biophys Acta 1992; 1122:143–6.


18. Liu X, Sun Q, Wang H, Zhang L, Wang JY. Microspheres of corn protein, zein, for an ivermectin drug delivery system. Biomaterials 2005; 26:109–15.


19. National Pharmacopoeia Commission. Pharmacopoeia of People’s Republic of China. Beijing: Chemical Industry Press; 2010.
20. Luo Y, Zhang B, Whent M, Yu LL, Wang Q. Preparation and characterization of zein/chitosan complex for encapsulation of α-tocopherol, and its in vitro controlled release study. Colloids Surf B 2011; 85:145–52.

21. Fernandes P. Enzymes in food processing: a condensed overview on strategies for better biocatalysts. Enzyme Res 2010; 2010:1–19.



22. Bassegoda A, Cesarini S, Diaz P. Lipase improvement: goals and strategies. Comput Struc Biotechnol J 2012; 2:1–8.



23. Tümtürk H, Karaca N, Demirel G, Sahin F. Preparation and application of poly (N, N-dimethylacrylamide-co-acrylamide) and poly (N-isopropylacrylamide-co-acrylamide)/κ-Carrageenan hydrogels for immobilization of lipase. Int J Biol Macromol 2007; 40:281–5.


24. Nawani N, Singh R, Kaur J. Immobilization and stability studies of a lipase from thermophilic Bacillus sp: The effect of process parameters on immobilization of enzyme. Electron J Biotechnol 2006; 9:559–65.

25. Kenthorai RJ, Chan ES, Pogaku R. Physical and stability characteristics of Burkholderia cepacia lipase encapsulated in κ-carrageenan. J Mol Catal B: Enzym 2009; 58:78–83.

26. Inborr J, Bedford MR. Stability of feed enzymes to steam pelleting during feed processing. Anim Feed Sci Technol 1994; 46:179–96.

27. Su DH, Liu P, Zheng YA, Sun JJ. Increased thermostability of phytase by coated conditions. Chinese J Bioprocess Eng 2004; 3:240–5.
- TOOLS
-
METRICS

- Related articles
-
In vitro evaluation of Rhus succedanea extracts for ruminants2018 October;31(10)
In vitro Arsanilic Acid Induction of Apoptosis in Rat Hepatocytes2006 September;19(9)
In vitro Evaluation of Phalaris minor Seeds as Livestock Feed2006 March;19(3)
Cow faeces in in vitro digestibility assays of forages1998 February;11(1)





 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC Download Citation
Download Citation Print
Print




