Effects of Dietary Scutellaria baicalensis Extract on Growth, Feed Utilization and Challenge Test of Olive Flounder (Paralichthys olivaceus)
Article information
Abstract
Effects of dietary Scutellaria baicalensis extract (SBE) on growth, feed utilization and challenge test of olive flounder (Paralichthys olivaceus) were determined. Six hundred thirty fish averaging 5.0 g were distributed into 18, 180-L tanks. Six experimental diets were prepared in triplicate: SBE-0, SBE-0.5, SBE-1, SBE-2, SBE-3 and SBE-5 diets containing SBE at the concentrations of 0, 0.5, 1, 2, 3 and 5%, respectively. Fish were hand-fed to apparent satiation twice a day for 8 wks. At the end of 8-wk feeding trial, ten fish from each tank were infected by Edwardsiella tarda for challenge test. Survival and weight gain of fish were not affected by dietary concentrations of SBE. However, specific growth rate of fish fed the SBE-2 diet was higher than that of fish fed the SBE-0, SBE-1, SBE-3 and SBE-5 diets. Neither feed efficiency nor serum chemical composition of fish was affected by dietary concentrations of SBE. The cumulative mortality of fish fed the SBE-0 diet was 100% at 96 h after E. tarda infection, but 77 to 87% for fish fed the other diets. Dietary inclusion of 2% SBE appears to be recommendable to improve specific growth rate of fish and SBE had the potential to mitigate mortality of fish at E. tarda infection.
INTRODUCTION
Olive flounder (Paralichthys olivaceus) is one of the most important marine finfish for aquaculture in Eastern Asia including Korea, Japan and China because of its fast growth and high resistance to disease. Therefore, many feeding trials to determine dietary nutrient requirements (Lee et al., 2000a, 2002; Kim and Lee, 2004), optimum feeding ratio (Cho et al., 2006c, 2007a), feeding frequency (Lee et al., 1999, 2000b), alternative protein sources for fishmeal in the diets (Kikuchi et al., 1994, 1997; Sato and Kikuchi, 1997; Kikuchi et al., 1999; Kim et al., 2000b), best feeding strategy (Kim et al., 2002a; Cho, 2005; Cho et al., 2006b) have been performed.
Dietary additives have been developed to improve survival, growth, immune resistance and/or muscle quality of olive flounder; herb, obosan™ (Kim et al., 1998, 2000a), lactic acid bacteria cultured in herb (Acanthopanax koreanum) extract (Jhon et al., 2009), extract of mushroom mycelium (Phellinus linteus and Coriolus militaris) (Kim et al., 2006), Chlorella powder (Kim et al., 2002b), green tea (Cho et al., 2006a, 2007b) and chitosan (Kim et al., 2005; Cha et al., 2008). Furthermore, dietary additives such as Chinese herb mixture (Astragalus radix and Ganoderma lucidum) for carp (Cyprinus carpio) (Yin et al., 2009) and Radix astragalin seu Hedysari and R. angelicae sinensis for yellow croaker (Pseudosciaena crocea) (Jian and Wu, 2003) and Indian herb (Ocimum sanctum Linn) for tilapia (Oreochromis mossambicus) (Logambal et al., 2000) were reported to improve survival after disease infection. Still, since mortality resulted from outbreak of disease in commercial flounder farm throughout year-round culture cause economic loss to fish farmer, development of a new additive to improve its disease resistance is highly desired.
The use of natural herb has the several advantages as a dietary additive, such as food safety of fish for human consumption. An extract of Scutellaria baicalensis, in which baicalin and baicalein are the major active components has been known to have antimicrobial, antibacterial, antiviral, antiinflammatory and/or antioxidant activities (Kim et al., 1999a, 2000c; Tan and Vanitha, 2004; Hwang et al., 2006) and recently developed as additive for aquafeed. Yin et al. (2006) also reported that dietary inclusion of 0.5 and 1% S. radix showed an inhibition of phagocytosis and respiratory burst activity in tilapia. Galina et al. (2009) reviewed the use of herbs as an immunostimulant in fish and demonstrated their effects on immune response and disease resistance.
In this study, therefore, effects dietary S. baicalensis extract on growth, feed utilization and challenge test of olive flounder were determined.
MATERIALS AND METHODS
Experimental conditions
Juvenile olive flounder were purchased from a private hatchery (Taean, Chungcheongnam Do, Korea), transferred into the laboratory and acclimated for 2 wks before an initiation of the feeding trial. During the acclimation period, fish were fed a commercial feed containing 54% crude protein and 11% crude lipid (Suhyup Feed Co. Ltd., Korea) twice a day at 2 to 3% total body weight. Six hundred thirty juvenile fish averaging 5.0 g were randomly distributed into 18, 180-L flow-through tanks (thirty five fish per tank) and water flow rate of each tank was 6.8-L/min. Each tank was aerated and water temperature ranged from 18.4°C to 24.1°C (mean±SD: 21.5±1.2°C).
Preparation of experimental diets
A commercially available extract of S. baicalensis extract (SBE) (Bioskintech Co. Ltd., Seoul, Korea) containing 100 ppm baicalin was used as a dietary additive. Six experimental diets were prepared in triplicate: SBE-0, which was used as control diet, SBE-0.5, SBE-1, SBE-2, SBE-3 and SBE-5 diets containing SBE at the concentrations of 0, 0.5, 1, 2, 3 and 5%, respectively (Table 1). The designated concentrations of SBE, which was an aqueous type, were included into the experimental diets instead of the same amount of water. Fishmeal, dehulled soybean meal and corn gluten were used as protein source for the experimental diets. Wheat flour, and fish and soybean oils were used as carbohydrate and lipid sources, respectively. The experimental diets were formulated based on the results of Yigit et al. (2004) and Lee and Kim (2005)’s studies showing that growth of olive flounder did not reach the plateau at the highest protein levels of 50 to 51% tested in their studies. The ingredients of the experimental diets were mixed well with water at a ratio of 3:1 and pelletized with a pellet-extruder. The experimental diets dried at room temperature overnight and stored at −20°C until use. All fish were hand-fed to apparent satiation twice a day (07:00 and 17:00 h), seven days a week, for 8 wks.
Chemical analysis of the experimental diets and biological index
Ten fish at the initiation and five fish from each tank at the termination of the feeding trial were sampled and sacrificed for biological index of fish. The experimental diets were analyzed for chemical analysis. Five fish from each tank were individually weighed, measured and then dissected to collect liver. HSI and CF were calculated as follows: Hepatosomatic index (HSI) = liver weight×100/fish weight, and condition factor (CF) = fish weight×100/total length3.
Serum analysis
At the end of the 8-wk feeding trial, blood samples were obtained from the caudal vein of three randomly chosen fish from each tank. Fish were starved for 24 h prior to bleeding. Serum was collected after centrifugation (900 g for 10 min), pooled by tank, stored freezer at −70°C as separate aliquots for analysis of total protein, glucose, glutamate oxaloacetate transaminase (GOT), glutamate pyruvate transaminase (GPT), and triglyceride, and analyzed by using automatic chemistry system (Vitros DT60 II, Vitros DTE II, DTSC II Chemistry System, Johnson and Johnson Clinical Diagnostics Inc., New York, USA).
Lysozyme activity assay
The turbidimetric assay for lysozyme was carried out according to Parry et al. (1965). Briefly, test serum (100 μl) was added to 2 ml of a suspension of Micrococcus lysodeikticus (0.2 mg/ml, Sigma) in a 0.05 M sodium phosphate buffer, pH 6.2. The reaction was carried out at 25°C and absorbance at 530 nm was measured on spectrophotometer after 0.5 and 4.5 min. A lysozyme activity unit was defined as the amount of enzyme producing a decrease in absorbance of 0.001/min.
Challenge test
Ten fish from each tank were chosen at the end of the 8-wk feeding trial and shown to be free from bacterial infection. Fish were held in the same tanks as the feeding trial without water exchange. The bacteria used for challenge test were obtained as a reference pathogenic strain of Edwardsiella tarda (FSW910410). A culture suspension of E. tarda was grown agar for 24 h, collected, washed and suspended in a sterile 0.85% saline solution and counted. Then, fish were artificially infected by intraperitoneal injection with 0.1 ml of culture suspension of pathogenic E. tarda containing 4×108 cells/ml. Fish were monitored for the next 96 h after E. tarda infection and dead fish were removed every 3 h for the first 24 h, 6 h for the second 24 h and 12 h for the last 48 h. Water temperature ranged from 22.3°C to 23.0°C.
Statistical analysis
One-way ANOVA and Duncan's multiple range test (Duncan, 1955) with SAS version 9.1 (SAS Institute, Cary, NC, USA) were used to analyze the significance of the difference among the means of treatments.
RESULTS
Survival of fish (over 98%) for all experimental diets and weight gain of fish ranged from 545% to 637% were not significantly (p>0.05) affected by dietary concentrations of SBE (Table 2). However, SGR of fish fed the SBE-2 diet was significantly (p<0.05) higher than that of fish fed the SBE-0, SBE-1, SBE-3 and SBE-5 diets, but not significantly (p>0.05) different from that of fish fed the SBE-0.5 diet.
Feed consumption of fish fed the SBE-2 diet was significantly (p<0.05) higher than that of fish fed the other diets (Table 3). Also feed consumption of fish fed the SBE-0 diet was significantly (p<0.05) higher than that of fish fed the SBE-5 diet, but not significantly (p>0.05) different from that of fish fed the SBE-0.5, SBE-1 and SBE-3 diets. Feed efficiency ratio (PER) and protein retention (PR) for olive flounder were not significantly (p>0.05) affected by dietary concentrations of SBE. However, Protein efficiency ratio (PER) for fish fed the SBE-5 diet was significantly (p<0.05) higher than that for fish fed the SBE-1, SBE-2 and SBE-3 diets, but not significantly (p>0.05) different from that for fish fed the SBE-0 and SBE-0.5 diets. HSI and CF of fish was not significantly (p>0.05) affected by dietary concentrations of SBE.
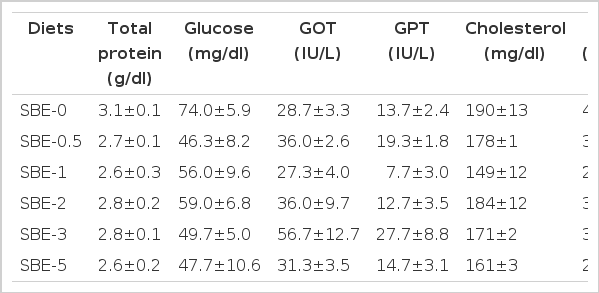
Serum total protein level ranged from 2.6 g/dl to 3.1 g/dl, glucose level ranged from 46.3 mg/dl to 74.0 mg/dl, GOT level ranged from 27.3 IU/L to 56.7 IU/L, GPT level ranged from 7.7 IU/L to 27.7 IU/L, cholesterol level ranged from 149 mg/dl to 190 mg/dl and triglyceride level ranged from 218 mg/dl to 459 mg/dl and none were significantly (p>0.05) affected by dietary concentrations of SBE (Table 4). No significant (p>0.05) difference in lysozyme activity of olive flounder fed the experimental diets with the various concentrations of SBE for 8 wks was observed.
Serum chemical composition and lysozyme activity of olive flounder at the end of the 8-wk feeding trial (n = 3)
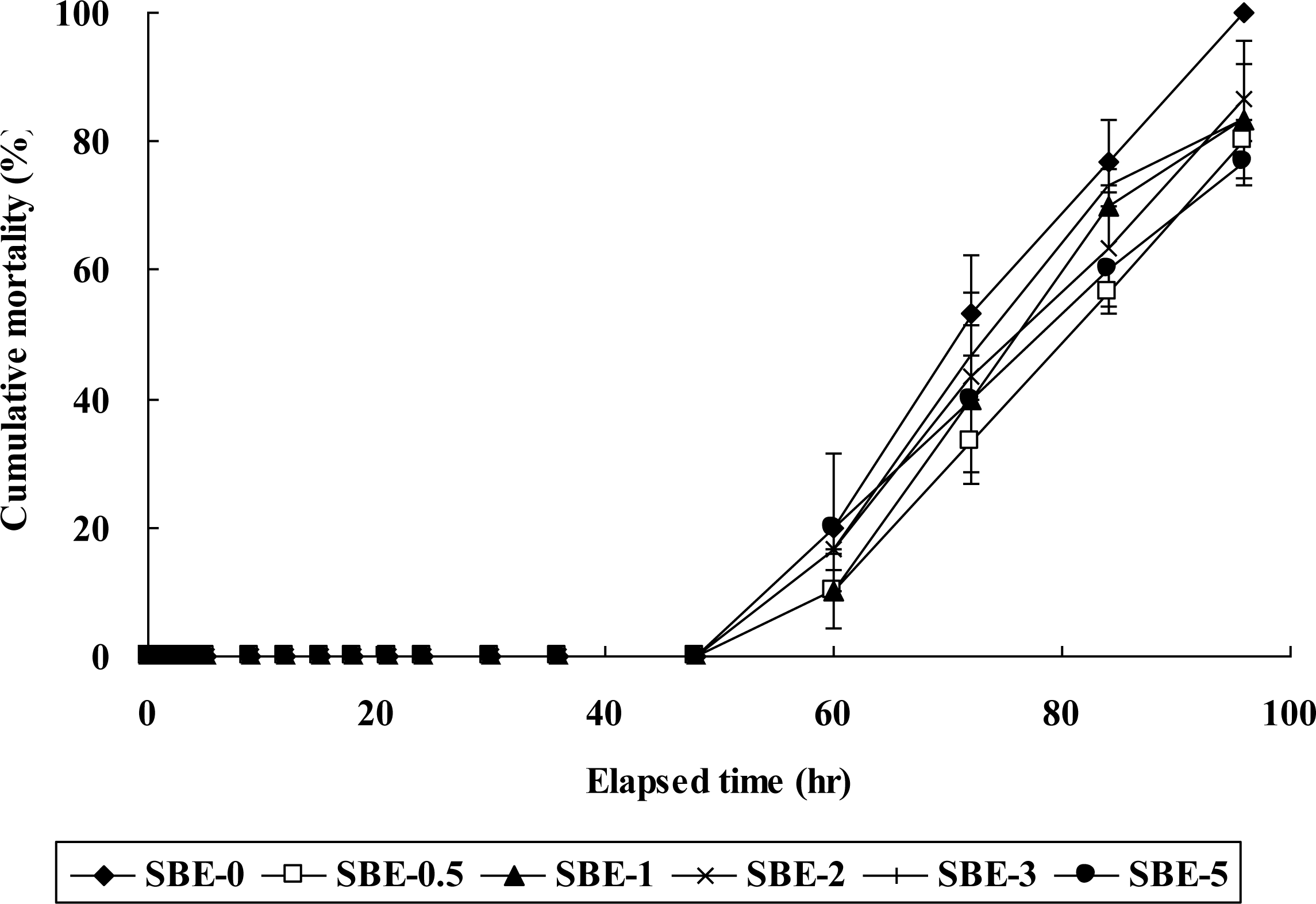
Mortality was initially observed in all groups at 60 h after E. tarda infection (Figure 1). Fish fed the SBE-0 diet showed 100% mortality at 96 h after E. tarda infection, but 77 to 87% for fish fed the other (SBE-0.5, SBE-1, SBE-2, SBE-3 and SBE-5) diets; however, no significant (p>0.05) difference was found among the experimental diets.
DISCUSSION
Use of herbal extracts as immunostimulants that enhance the innate (non-specific) immune response in fish have been reviewed (Galina et al., 2009). The major components of the innate immune system of fish were macrophages, monocytes, granulocytes and humoral elements, such as lysozyme or the complement system (Sakai, 1999). Herbs are commonly used in commercial aquaculture as a growth-promoting substance due to their food safety for human consumption. Similarly, the growth-promoting effect was observed in fish fed the SBE-2 diet because SGR of fish fed the SBE-2 diet was higher than that of fish fed the other diets (SBE-0, SBE-1, SBE-3 and SBE-5 diets) in this study. Obosan™ which is one of the most commonly used herbs by olive flounder farmers in Korea effectively improved not only weight gain of olive flounder (Kim et al., 1998, 2000a), but also non-specific immune response (lysozyme activity and nitroblue tetrazolium reduction of macrophage) (Park et al., 2003). In addition, dietary inclusion of extract of mushroom mycelium P. linteus and C. militaris (Kim et al., 2006) and C. ellipsoidea powder (Kim et al., 2002b) improved weight gain of olive flounder. However, unlike these studies, dietary supplementation of chitin and chitosan depressed growth of hybrid tilapia (O. niloticus×O. aureus) (Shiau and Yu, 1999). Therefore, application of dietary additives should be carefully considered because their favorable roles in fish vary depending on fish species, targeting activities of additives, administration method, dose (concentration), physiological and nutritional status of fish and/or dietary nutrition composition, etc.
FER and PR for olive flounder were not significantly affected by dietary concentrations of SBE in this study. However, feed efficiency of fish improved with dietary inclusion of additives resulting from an increased weight gain of fish (Kim et al., 1998, 2000a, 2002b; Jhon et al., 2009; Seo et al., 2009).
None of serum total protein, glucose, GOT, GPT, cholesterol and triglyceride level were significantly affected by dietary concentrations of SBE in this study. Park et al. (2003) observed similar effects that dietary inclusion of additives did not affect fish serum criteria. However, others have shown that dietary inclusion dietary inclusion of extract of mushroom mycelium, green tea, and lactic acid bacteria cultured in herb extract lowered serum GPT, GPT and low density lipoprotein cholesterol, and GOT and GPT of olive flounder, respectively (Kim et al., 2006; Cho et al., 2007b; Jhon et al., 2009) although the mechanism had not been elucidated yet. Bok et al. (1999) reported that administration of citrus bioflavonoid lowered the plasma cholesterol concentration and fecal neutral sterol in high cholesterol-fed rats. Previously, dietary inclusion of obosan™ increased serum total protein and glucose contents, but lowered GOT and GPT of fish (Kim et al., 2000a). One explanation for these differences may be the various sources of additives.
No difference in lysozyme activity of olive flounder fed the experimental diets with the various concentrations of SBE in this study was in agreement with Yin et al. (2006) study showing that administration of 0.1, 0.5 and 1% S. radix did not affect lysozeme activity of tilapia, but 0.1 and 0.5% Astragalus radix increased it. Administration of Chinese herb mixture (A. radix and G. lucidum) stimulated respiratory burst activity, phagocytosis of phagocytic cells and lysozyme activity of carp (Yin et al., 2009). In addition, Indian herb (Solanum trilobatum) elevated lysozyme activity of tilapia (Divyagnaneswari et al., 2007). Lysozyme is part of the nonspecific immune system (Jolles and Jolles, 1984) and its activity level is affected by stress although the response following acute or prolonged stress is ambiguous (Roed et al., 1993; Demers and Bayne, 1997). Moreover, Roed et al. (2002) demonstrated that the use of lysozyme activity as a selection criterion to improve disease resistance was complicated by the fact that the association between lysozyme activity and disease resistance appeared to be considerably influenced by the immune status of the fish at timing of sampling blood for lysozyme assessments in rainbow trout (Oncorhynchus mykiss).
No significant difference in cumulative mortality of fish fed the experimental diets with various concentration of SBE was found in this study. However, a trend toward into a decreased cumulative mortality of fish fed the diets with SBE probably indicates that S. baicalensis extract can have the potential as dietary additive to mitigate mortality of fish infected with E. tarda. However, pathogen concentration (4×108 cells/ml E. tarda) used for fish infection in this study seemed to be too high because high mortality was obtained in all fish groups.
Similarly, administration of Chinese herb mixture (A. radix and G. lucidum) for carp and Indian herb (O. sanctum or Solanum trilobatum) for tilapia improved immune response and disease resistance against Aeromonas hydrophia (Logambal et al., 2000; Divyagnaneswari et al., 2007; Yin et al., 2009). Also, dietary inclusion of Chinese herb mixture (R. astragalin seu Hedysari and R. angelicae sinensis) (Jian and Wu, 2003) and aloe (Kim et al., 1999b) elevated survival of fish compared to control fish, following a challenge with V. alginolyticus. Dietary inclusion of extract of mushroom mycelium (P. linteus and C. militaris) (Kim et al., 2006), green tea (Cho et al., 2006) and Undaria (Kwon et al., 2003) improved survival of fish after E. tarda infection. In addition, dietary inclusion of lactic acid bacteria cultured in herb (A. koreanum) extract improved survival of fish after either Vibrio anguillarum or Streptococcus iniae infection (Jhon et al., 2009). Chitosan reduced mortality of olive flounder by 36.5% on commercial farm for 12-month feeding trial (Kim et al., 2005).
Results of this study indicates that dietary inclusion of 2% S. baicalensis extract seems to be recommendable to improve SGR of olive flounder and S. baicalensis extract have the potential as an additive to mitigate mortality of fish at E. tarda infection.
Acknowledgements
This research was a part of the project titled “Development of Technologies for Risk Assessment of Marine Living Genetically Modified Organisms” funded by the Ministry of Land, Transport and Maritime Affairs, Korea and a grant funded by Ministry of Maritime Affairs and Fisheries of Korean Government (F20816608H220000110). In addition, I appreciate Dr. Kwon, Mun-Gyeong at National Fisheries Research and Development Institute (Busan, Korea) for performing disease challenge test