Effect of Dietary Supplementation of Procyanidin on Growth Performance and Immune Response in Pigs
Article information
Abstract
This study was performed to determine the effect of dietary supplementation of procyanidin on growth performance, blood characteristics, and immune function in growing pigs. In experiment 1 (Exp. 1), thirty-two crossbred pigs with an initial BW of 19.2±0.3 kg were allocated into 4 treatments for an 8-wk experiment: i) CON (basal diet), ii) MOS 0.1 (basal diet+0.1% mannanoligosaccharide), iii) Pro-1 (basal diet+0.01% procyanidin), and iv) Pro-2 (basal diet+0.02% procyanidin). Pigs fed Pro-1 and Pro-2 diets had greater (p<0.05) gain:feed ratio compared with those fed CON or MOS 0.1 diets. Serum creatinine concentration was less (p<0.05) in Pro-2 treatment than those in CON, MOS 0.1 and Pro-1 treatments. In Exp. 2, twelve pigs (BW 13.4±1.3 kg) received basal diet with i) 0 (CON), ii) 0.02% (Pro-0.02%), and iii) 0.04% procyanidin (Pro-0.04%) for 4 wk. Concentration of platelets was lower (p<0.05) in the Pro-0.04% group compared to CON at 24 h after lipopolysaccharide (LPS) challenge. In addition, secretion of cytokines from cultured peripheral blood mononuclear cells (PBMC) in the presence or absence of procyanidin was examined. The levels of interleukin (IL)-1β, IL-6 and tumor necrosis factor (TNF)-α were lower (p<0.05) in Pro (LPS-stimulated PBMCs+procyanidin) than those in CON (LPS-stimulated PBMCs+PBS) at 4 h after LPS challenge. These data suggest that dietary addition of procyanidin improves feed efficiency and anti-inflammatory cytokines of pigs.
INTRODUCTION
During the past decades, antibiotics have been used as feed additives to improve growth of animals and protect them from pathogenic microorganisms (Meng et al., 2010). However, in recent years, public concerns over the use of antibiotics in livestock have increased due to the emergence of antibiotic-resistant bacteria that may be a hazard to human health (Huang et al., 2010). Therefore, European Union (EU) started to ban the use of antibiotic as feed additives in 1999 and now are considering restricting its use outside EU (Windisch et al., 2008). In Korea, it has been required to develop antibiotic-free diets for domestic animals including pigs (Bae et al., 1999). Accordingly, considerable effort has been dedicated to identifying alternatives to antibiotics as growth promoters in the animal industry (Hong et al., 2004; Cho et al., 2006).
Procyanidins have been reported to exert antioxidant properties (Balu et al., 2005; Mohanasundari et al., 2005; Sangeetha et al., 2005; Yahara et al., 2005). They are present in the grape seed extracts and are composed of flavan units linked by C4-C6 or C4-C8 bonds forming dimmers, trimers, tetramers, and polymers up to 15 to 16 units (Prieur et al., 1994) with small amounts of catechin and epicatechin (Ricardo da Silva et al., 1990). Flavan compounds possessed many biological effects including scavenging free radicals, chelating transition metals, and modulating activities of antioxidant enzymes. Therefore, they have anti-aging effect, alleviate bone debilities, and protect renal tissues against toxicity. Procyanidins are also known to have therapeutic effects with anti-mutagenic (Faria et al., 2006), anti-cytotoxic (Cos et al., 2004), and anti-inflammatory (Selmi et al., 2006) activities. Therefore, this study was performed to determine the effect of oral administration of procyanidin on growth performance and immune response in growing pigs.
MATERIALS AND METHODS
Preparation of procyanidin
Procyanidin supplied by SINE-BIO (Seongnam, Korea) was collected and sterilized as described by Hwang et al. (2011). Grape seeds were powdered and soaked in a mixture of acetone/water/acetic acid (v/v/v 70:29.5:0.5). The mixture was filtered and dried under reduced pressure using a rotary evaporator at 40°C. It was further dried in a vacuum-freeze dryer and 70% acetone extracts were obtained. Extracts were then separated by n-hexane, and separated by an open column chromatography packed with silica gel. The stationary phase was sephadex LH-20 and the mobile phase consisted of a mixture of methanol and water (v/v 20:80). The flow rate was 1.2 mL/min and the sample was analyzed at 283 nm with a spectrometer. The fraction with a retention time of 18.7 min (89% purity) showed the highest activity and 18 mg of the procyanidin extract was collected from this fraction. Finally, it was dried using a freeze drier. Concentration of extracted procyanidin used in this experiment was mixed with feed at 0.265%.
Animal managements and experimental diets
The experimental protocols describing the management and care of animals were reviewed and approved by the Animal Care and Use at the National Institute of Animal Science (NIAS). All diets (Table 1) were formulated to meet the nutrient requirements recommended by the official Korean Feeding Standard for Swine (NIAS, 2007). Each pen was provided with a stainless steel feeder and one nipple drinker that allowed for ad libitum access to feed and water throughout the experiment.
Experiment 1: feeding trial
Experimental design and animals
A total of 32 barrows (Landrace×Yorkshire) with an average initial BW of 19.2±0.3 kg were selected for this 8 wk growth trial. Pigs were allocated to 1 of 4 dietary treatments according to their BW in a randomized complete block design with 8 pigs per treatment group. Dietary treatments included i) CON (basal diet), ii) MOS 0.1 (basal diet+0.1% mannanoligosaccharide), iii) Pro-1 (basal diet+0.01% procyanidin), and iv) Pro-2 (basal diet+0.02% procyanidin).
Growth performance measurements, blood sampling and analysis
Individual pig BW and feed consumption were recorded on d 0 and at the end of the experimental period on d 56 and ADG, ADFI, and gain:feed (G:F) were calculated. Blood samples (10 mL) were collected via anterior vena cava puncture after the pigs had been starved for 12 h at the beginning, 3 wk, and 8 wk of experimental period. Whole blood sample was centrifuged at 2,000×g for 10 min at 4°C, and the serum was collected. Serum biochemical profile was determined using a chemistry analyzer (COBAS MIRA plus, ROCHE diagnostics. Block Scientific, Inc. Bohemia, NY, USA).
Superoxide dismutase (SOD) activity
In our study, SOD was measured to investigate the effect of in vivo antioxidant activity of hesperidin. The SOD was determined by the method of Marklund and Marklund (1974). In a cap tube containing 100 μL of serum sample (9 wk serum), 1.5 mL of Tris EDTA HCl buffer (50 mM Tris, 1 mM EDTA, 6 N HCl) with 50 μL of pyrogallol (5 mM) was incubated at 25°C for 30 min. Thereafter, 50 μL of 1 N HCl was added and vortexed for several seconds. The absorbance of the solution was measured at 420 nm. One unit was the amount of enzyme required to cause 50% inhibition of pyrogallol oxidation, and SOD activity was expressed as unit/mg protein.
Experiment 2: Lipopolysaccharide (LPS) challenge trial
Experimental design and animals
A total of 12 barrows (Landrace×Yorkshire) with an average initial BW of 13.4±1.3 kg maintained in individual pens were assigned to 3 dietary treatments in a randomized complete block design based on BW for a 4-wk period. Three treatments included: i) CON (basal diet), ii) Pro-0.02% (basal diet+0.02% procyanidin), and iii) Pro-0.04% (basal diet+0.04% procyanidin). The level of procyanidin was increased to 0.04% since pigs needed to have more procyanidin under LPS challenge to give rise to the effect of procyanidin on pro-inflammatory cytokines. At the end of 4 wk, all pigs in each dietary treatment were intraperitoneally injected with Escherichia coli (serotype 0111:B4) LPS (Sigma Chemical Co., St, Louis, MO, USA) at a level of 50 μg/kg of BW. The LPS dosage was referenced on the results of previous studies (Matteri et al., 1998; Wright et al., 2000; Kim et al., 2010).
Blood sampling and measurements
Blood samples (15 mL) were collected via anterior vena cava puncture at 0 and 24 h after LPS injection after the pigs had been starved for 12 h. Blood samples were collected into both non-heparinized tubes (10 mL) and vacuum tubes (5 mL) containing K2 EDTA (Becton, Dickinson and Co., Franklin Lakes, NJ, USA) to obtain serum and whole blood, respectively. Blood samples were centrifuged (2,000×g) for 10 min at 4°C. The white blood cells, red blood cells and platelet concentration in the whole blood were determined using an automatic blood analyzer (Hemavet 950, CDC. Drew Scientific Inc. Dallas, TX, USA). Serum immunoglobulin A (IgA), G (IgG) and M (IgM) concentrations were evaluated using commercially available ELISA Starter Accessory kits (Bethyl Laboratories, Inc. Montgomery, TX, USA). The absorbance was read at 450 nm using a microplate reader (Versamax, Molecular Devices, Inc. Sunnyvale, CA, USA).
Peripheral blood mononuclear cells (PBMCs) culture and cytokine secretion
Peripheral blood samples were collected in heparin-containing tubes (Becton, Dickinson and Co., Franklin Lakes, NJ, USA) from 2 healthy piglets that were not used in the experiments. Blood samples were diluted with PBS and layered over a Ficoll-Hypaque (GE Healthcare Bio-Sciences AB. Rapsgatan 7, Uppsala, Sweden) gradient and centrifuged at 2,200×g for 17 min at 10°C. PBMCs were harvested from the interface layer, washed twice with PBS and then counted. Cells were resuspended in RPMI (Roswell Park Memorial Institute) 1640 GlutaMax (Invitrogen, GIBCO. Carlsbad, CA, USA). Concentration of PBMC was adjusted to 1.25×106 viable cells/mL after estimation of viability by trypan blue exclusion assay. For the cytokine secretion assay, 200 μL of a 1.25×106 cell suspension was cultured with an equal volume of flavonoid 25 μg/mL (procyanidin 25 μg/mL) or PBS. We set the concentration of flavonoids used in this study based on prior dose response analysis conducted by our laboratory (Sanbongi et al., 1997; Mao et al., 1999).
Isolated PBMCs were stimulated with 100 ng/mL LPS and then cultured in addition of PBS (CON) or procyanidin (Pro) for 2 h or 4 h at 38°C with 5% CO2. PBMC interleukin (IL)-1β, IL-6 and tumor necrosis factor (TNF)-α concentrations were evaluated using commercially available ELISA kits (Quantikine, R&D systems, Minneapolis, MN, USA) and the absorbance was measured at 450 nm.
Statistical analysis
In this experiment, data were analyzed by using GLM procedure of SAS (SAS Institute, 2008). Differences among treatments were analyzed by Duncan’s multiple range test (Duncan, 1955). Probability values less than 0.05 were considered significant. Each pen was considered as an experimental unit. In Exp. 2, Cytokine secretion data were analyzed using t-test of SAS (SAS Institute, 2008). Each animal was considered as an experimental unit. Probability values less than 0.05 were considered significant.
RESULTS
Experiment 1: feeding trial
Growth performance
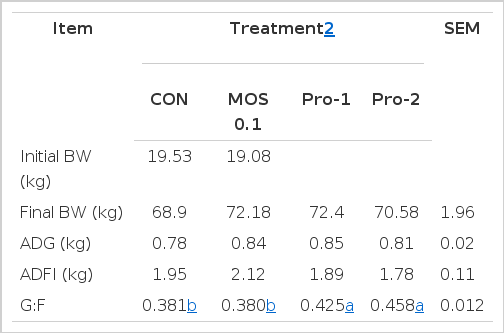
During the overall period, no difference was observed in BW, ADG, and ADFI among treatments (Table 2). However, dietary Pro-1 (0.425) and Pro-2 (0.458) treatments increased (p<0.05) G:F compared to CON (0.381) or MOS 0.1 (0.380) group.
Serum biochemical profile
The effects of procyanidin supplementation on serum profile in growing pigs are shown in Table 3. No effects were observed on cholesterol, total protein, albumin and blood urea nitrogen among treatments. However, serum creatinine concentration was significantly lower (p<0.05) in Pro-2 treatment than those in CON, MOS 0.1 and Pro-1 treatments. The glutamic oxalate transaminase concentration was greater (p<0.05) in MOS 0.1 treatment than that in CON treatment on 8 wk. Concentration of lactate dehydrogenase was lower (p<0.05) in CON treatment than all other groups at 3 wk.
Experiment 2: LPS challenge trial
Blood characteristics
White blood cell, red blood cell, hemoglobin, hematocrit, immunoglobulin A, G, and M were not influenced by dietary treatments (Table 5). Pigs administrated with Pro-0.04% had lower (p<0.05) platelet concentrations at 24 h after LPS injection compared to control group.
Cytokine secretion following LPS activation
Secretion of IL-1β and TNF-α were lower (p<0.05) in Pro (LPS-stimulated PBMCs+procyanidin) treatment than those in CON (LPS-stimulated PBMCs+PBS) treatment at 2 h and 4 h after LPS challenge, and IL-6 was lower (p<0.05) in Pro treatment than in CON treatment at 4 h after LPS challenge (Table 6).
DISCUSSION
Experiment 1: feeding trial
Growth performance
Endotoxin activity of procyanidin was tested by using Lumulus amebocyte lysate (LAL) and no detectable endotoxin was found (Holderness et al., 2007). Procyanidin’s endotoxin free property was again tested in various in vivo and in vitro studies including many animals and cell types (Bentivegna, 2002; Yamakoshi et al., 2002; Daughenbaugh et al., 2011; Lluis et al., 2011). Concomitant with these results, pigs treated with very low level of procyanidin, which was compared to those used in toxicity studies, had no sign of harmful effects on health or pro-inflammatory cytokines.
Discrepancies have been found in the effect of procyanidin on feed intake or growth performance that are fully or partly related to the differences in level of procyanidin in the experimental diets or in animal species used. Supplementation of antioxidant improves growth performance and feed efficiency (Lohakare et al., 2005). Previous studies reported that procyanidin helps reduce stress-related problems and improve growth rate in humans (Wooden et al., 1984; Slayback and Ronald, 2006). Procyanidin also showed these beneficial effects in rabbits (Garcia et al., 2002) and broiler chicks (Brenes et al., 2010). Concomitant with these data, results from the present study showed that administration of procyanidin increased the G:F ratio (Table 2) as well as the antioxidant activity in growing pigs (Table 4). Its beneficial effects on inflammation could reduce energy needs for immune responses thereby maintain animal health and improve growth performance. Therefore, it is possible that procyanidin be used as a feed additive for improving animal health and productivity.
Serum biochemical profile
Blood urea nitrogen (BUN) and serum creatinine (Scr) are well-known indicators of the renal function and health (Chen et al., 2003). However, Scr is a more specific indicator than BUN, since level of BUN can be affected by protein intake and liver function (Mathieson, 2003). Shi et al. (2003) reported that the antioxidant effect of procyanidin is approximately 50 times greater than that of vitamin C or vitamin E. Procyanidin alleviates the gentamicin induced-kidney injury (Jeong et al., 2005; Han et al., 2008; Hwang et al., 2008; Safa et al., 2010). Animal experiments have also demonstrated that procyanidin decreases the Scr concentrations in mice and rats (Kalantari et al., 2007; Yanarates et al., 2008). Concentration of Scr was also decreased by procyanidin treatment in our study (Table 3), indicating that procyanidin may have beneficial effects on renal health. However, it will be important to identify the relationship between procyanidin and serum creatinine level and how it helps improve renal function.
Superoxide dismutase (SOD) activity
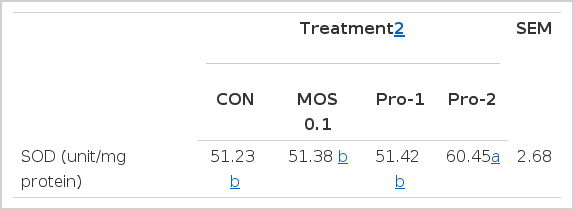
Superoxide is formed in the red blood cells by auto-oxidation of hemoglobin into methemoglobin (Nordberg and Arnér, 2001). Khan (1999) reported that SOD and glutathione peroxidase (GPX) have roles in an antioxidant defense system. These enzymes are involved in the clearance of superoxide and H2O2 to maintain the structure and function of biological membranes (McCord, 2000). Oguntibeju et al. (2012) reported that Wistar rats administered with procyanidin (0.87 mg/kg BW) showed increased SOD level compared to controls. Dietary supplementation of procyanidin also improved (p<0.05) the SOD-like activity in growing pigs from the present study (Table 4). This result is consistent with our hypothesis that supplementation of procyanidin may result in an increase in plasma flavanol level and an increase in the antioxidant capacity of plasma.
Experiment 2: LPS challenge trial
Blood characteristics
As a constituent of gram-negative bacteria cell walls, LPS induces the expression of many genes necessary for immune defense function (Tracey et al., 1994). Therefore, it is plausible to detect the inhibitory effect of procyanidin on the expression of inflammatory cytokines with a LPS challenge. Consequently, the present results confirm its ability to inhibit the production of multiple cytokines and thereby improve anti-inflammatory effect in pigs. Procyanidin has positive effects on the platelet concentration after LPS challenge (Zhang et al., 2006; Martinez-Micaelo et al., 2012). Zhang et al. (2009) reported that LPS promotes platelet activity by inducing the secretion of both α and dense granules, thus amplifying secretion-dependent platelet aggregation. Previous studies showed that platelets are involved in the pathogenesis of severe sepsis (Taylor et al., 1997; Pu et al., 2001; Zhao et al., 2002), and that LPS stimulates thrombosis, the formation of platelet microaggregates (Rumbaut et al., 2006).
Procyanidins are polymeric compounds derived from catechin and epicatechin (Pasinetti et al., 2010). They modulate platelet function, thus reduced the risk of clot formation (Karen et al., 2003). Pignatelli et al. (2000) suggested that procyanidin decreases platelet production induced by hydrogen peroxide. Procyanidin interferes oxidation process, decreases platelet activation and increases eicosanoid synthesis (Karen et al., 2003). These responses result in lowering the platelet activation, thereby decreases the concentration of platelets. Procyanidin also increases concentration of plasma prostacyclin. Prostacyclin decreases platelet aggregation in vivo and ex vivo by elevating platelet cyclic AMP concentration and inhibiting agonist-induced increase in glycoprotein IIb-IIa expression and phosphorylation. Administration of procyanidin inhibits platelet aggregation in rats (Chang and Hsu, 1989; Ruf et al., 1995), dogs (Demrow et al., 1995) and humans (Rein et al., 2000; Freedman et al., 2001). Concomitant with these results, pigs administrated with Pro-0.04% treatment also showed lower (p<0.05) platelet concentration compared to control group in this study (Table 5), suggesting that administration of procyanidin derived from catechin and epicatechin inhibits platelet aggregation in LPS-challenged pigs.
Cytokine Secretion Following LPS Activation
Inflammation and acute phase responses after LPS challenge have been well characterized in a pig model (Johnson, 1997; Wright et al., 2000). Several pro-inflammatory cytokines, such as TNF-α, IL-6, and IL-1, are encoded by target genes in the NFkB activation pathway (Yang et al., 2008). NFkB acts as a crucial transcriptional activator of pro-inflammatory cytokines and its expression is triggered by pro-inflammatory stimuli and genotoxic stress, including bacterial cell-wall components (Karin and Greten, 2005).
IL-1β, a pro-inflammatory cytokine, is a mediator of LPS toxicity in vivo and in vitro (Lee, 2007). Mao et al. (2005) reported that IL-1β level was increased by LPS, but pigs fed Astragalus membranaceus β-glucan (AMG) had a lower level of IL-1β than pigs from control group when they were challenged with LPS. TNF-α is produced at early onset of inflammation followed by waves of IL-1 and IL-6 (Tizard, 2000). However, TNF-α causes a detrimental effect on animal performance by compromising immunity and nutrient metabolism (Spurlock, 1997). These results may highlight the important role of these cytokines in regulation of immune and inflammatory responses. Procyanidin decreases the expression of IL-6 and TNF-α (Mackenzie et al., 2004; Erlejman et al., 2008; Terra et al., 2009). The potent anti-inflammatory property of procyanidin has been shown in rats and mice (Li et al., 2001; Sakaguchi et al., 2006). In this study, we assessed serum concentration of cytokines in association with dietary administration of procyanidin with experimentally imposed stimulation of the immune system by LPS. As expected, procyanidin decreased LPS-induced IL-1β, IL-6 and TNF-α production in PBMCs. Our results indicate that procyanidin may have the potential to suppress the inflammatory response and be effective for ameliorating the bacteria challenged anti-inflammatory response in pigs.
CONCLUSION
Taken together, dietary supplementation of procyanidin may help prevent over-stimulation of the immune system in growing pigs after an immunological challenge. In addition, procyanidin improves feed efficiency and decreases the serum creatinine concentrations in pigs. Further research, however, is necessary to identify the specific mechanisms by which procyanidin improves immune function and growth performance in growing pigs.
ACKNOWLEDGEMENTS
This work was carried out with the support of “cooperative research program for Agriculture Science & Technology Development (Project No. PJ009340)”, Rural Development Administration, Republic of Korea.