Effect of High Dietary Carbohydrate on the Growth Performance and Physiological Responses of Juvenile Wuchang Bream, Megalobrama amblycephala
Article information
Abstract
An optimum dietary carbohydrate content is important for maximum fish growth. In this study, we fed Wuchang bream (Megalobrama amblycephala) with either control diet (30.42%) or high carbohydrate diet (52.92%) for 90 d.
Fish were fed to apparent satiation three times daily in an aquarium with automatic temperature control and circulated water. Growth performance, plasma biochemical parameters, hepatic morphology and enzyme activities were determined. It was shown that compared to fish fed control diet, fish fed high carbohydrate diet had higher plasma triglyceride and cortisol levels for d 90, and lower alkaline phosphatase level for d 45, lower hepatic superoxide dismutase and total antioxidative capacity for d 90, higher malondialdehyde for d 45 and glycogen content for d 45 and 90 (p<0.05). Histological and transmission electron microscopy studies showed that hepatocytes of fish fed high carbohydrate diet contained large lipid droplets, causing displacement of cellular organelles to periphery of hepatocytes. The relative level of hepatic heat shock protein 70 (HSP70) mRNA of Wuchang bream fed high carbohydrate diet was significantly higher than that of fish fed the control diet for 90 d (p<0.05). These changes led to decreased specific growth rate and increased feed conversion ratio (p<0.05). Upon hypoxia challenge, fish fed high carbohydrate diet had higher cumulative mortality than those fed the control diet (p<0.05). These results suggested that high dietary carbohydrate (52.92%) was detrimental to the growth performance and health of Wuchang bream.
INTRODUCTION
Wuchang bream (Megalobrama amblycephala) is a Chinese freshwater herbivorous species (Ke, 1975) with high potential for aquaculture. This fish was originally found in Newshan Lake and Yuli Lake. Its main distribution is in the mid reach of the Yangtze River, China (Zhu, 1995). Due to the merits of this species including tender flesh, fast growth, feeding on natural foods, high disease resistance, economic profitability, and cultural values (Ke, 1986; Zhou et al., 2008; Li et al., 2010), Wuchang bream is widely cultured in China with the output of 625,789 ton in 2009; an increase of 31.50% in the past decade. This species has been introduced to North America (northern Canada to southern Mexico), Africa, Europe and other Asian countries. It can adapt well to local environment, being compatible with native species, and thus has a bright future for culture worldwide (Ke, 1986). According to previous reports (Zou et al., 1987; Shi et al., 1988; Yang et al., 1989; Liu et al., 1992; Zhou et al., 1997; Li et al., 2010), the optimum protein requirement of Wuchang bream ranges between 21.05 and 41.4% and the carbohydrate requirement was reported to be between 25% and 30%. However, little research has been done on the health implications of high dietary carbohydrate on Wuchang bream.
The carbohydrate utilization of fish varies between fish species and carbohydrate sources (Wilson, 1994; Krogdahl et al., 2005). Fish are known to have a limited ability for digestion and metabolism of carbohydrate and hence, excessive intake of this nutrient may result in nutritional problems (Jauncey, 1982; Roberts and Bullock, 1989; Lall, 1991). Excess carbohydrates reduce the growth rate and are often accompanied by poor feed utilization (Hemre et al., 2002). In general, herbivorous fish are more capable of utilizing dietary carbohydrate than carnivorous and omnivorous fish (Hemre et al., 2002). Fish with excess glucose are assumed to be under constant metabolic stress (Pieper and Pfeffer, 1980; Fletcher, 1981), which may lead to suppressed immune functions (Erfanullah, 1995; Fu and Xie, 2005) and cause a change in heat shock protein 70 (HSP70) content in gilthead sea bream Sparus aurata (Enes et al., 2006; Miao et al., 2011). Thus, high dietary carbohydrate consumption may increase the incidence of diseases and affect the antioxidation ability of the fish.
The structural modifications of nuclei observed within hepatocytes could reflect a nutritional pathology (Caballero et al., 1999). Histological changes in tissues of fish fed diets containing different carbohydrate levels have also been reported for some fish species. For instance, hepatocytes with large lipid vacuoles and nuclei located at the periphery of the cell were observed in livers of Labeo rohita juveniles (Kumar et al., 2005) fed Non-gelatinised carbohydrate and L. rohita fry (Mohapatra et al., 2003) fed higher gelatinized carbohydrate. Although the histopathological interpretation of these ultrastructural changes is difficult, it can provide useful information on better understanding the metabolism of carbohydrate.
Therefore, the objectives of the present study were to evaluate the effect of high dietary carbohydrate on growth performance, plasma biochemical indices, hepatic antioxidation enzyme activities and expression of hepatic HSP70 to gain a better understanding of the health implications of high dietary carbohydrate in Wuchang bream.
MATERIALS AND METHODS
Experimental diets
Two isonitrogenous (28.56% crude protein) and isolipidic (5.28% crude lipid) semi-purified diets were formulated to contain either 30.42% or 52.92% carbohydrate (Table 1). Fish meal was used as the only protein source, and α-starch (Wuxi Jinling Tower Starch Co. Ltd., China) as the only carbohydrate source. Dietary ingredients were ground through a 60-mesh screen. Vitamins and minerals were mixed by the progressive enlargement method (Zhou et al., 2007). Lipid and distilled water (60%, w/w) were added to the premixed dry ingredients and thoroughly mixed until homogenous in a feed mixer. The 2-mm diameter pellets were wet-extruded, air-dried, and sealed in plastic bags and stored frozen at −20°C until being used.
Animal rearing and feeding trial
Juvenile Wuchang bream (Megalobrama amblycephala) were obtained from the Freshwater Fisheries Research Center, Chinese Academy of Fishery Sciences, China. The fish were placed in six 300 L cylindrical fiberglass tanks and acclimated for 15 d. After the acclimation, fish of similar sizes (average weight, 34.73±0.67 g) were randomly distributed into six 300 L cylindrical fiberglass tanks at a rate of 28 fish per tank. Fish in each tank were randomly assigned one of two experimental diets. Each diet was tested in triplicate. Fish were hand-fed to apparent satiation three times daily (06:30, 11:30 and 16:30 h) for 90 d.
Fish were provided with a continuous flow of sand-filtered freshwater (2 L/min) with continuous aeration. Water quality parameters were monitored daily. During the feeding trial, water temperature was 26.0±1.5°C. The water oxygen content was not less than 6.0 mg/L during the experiment. A 12 L:12 D photoperiod was used. Tanks were thoroughly cleaned biweekly.
Sample collection
At the start of the experiment, the fish were weighed after 24-h starvation and nine fish from each group were sampled. The fish were anesthetized in diluted MS-222 (tricaine methanesulfonate, Sigma, USA) at the concentration of 200 mg/L before being sampled. Nine fish from each experimental group in the middle of the experiment were sampled again. After 24-h starvation at the end of the feeding period, fish from each tank were weighed and the specific growth rate (SGR) was calculated. Blood was collected from the caudal vein, immediately centrifuged and the plasma frozen for analysis. The livers were frozen in liquid nitrogen and stored at −80°C until assay.
Proximate analysis of Wuchang bream
Diets and fish were analyzed for proximate composition. Dry matter was determined by oven drying until constant weight (105°C). Crude protein (nitrogen×6.25) was determined by the Kjeldahl method using an Auto Kjeldahl System (FOSS KT260, Switzerland). Crude lipid was determined by ether-extraction using Soxtec System HT (Soxtec System HT6, Tecator, Sweden) and ash was determined by combustion at 550°C for 4 h. The hepatic glycogen content was determined as described in Plummer (1987).
Plasma biochemical parameters and hormones
Plasma triglyceride (TGL), glucose (GLU) and cholesterol (CHOL) were measured by enzymatic (glycerol phosphate oxidase) and colorimetric (PAP) methods, glucose oxidase method, and enzymatic (cholesterol oxydase) and colorimetric methods, respectively, using kits purchased from Junshi Biotechnology Co., Ltd., Shanghai, China. Plasma insulin was measured by radioimmunoassay (RIA) using a test kit (Beifang Biotech Research Institute, Beijing, China) and following the method described by Clark and Hales (Clark and Hales, 1994; Li et al., 2012). Plasma cortisol was measured by RIA using a test kit (Beifang Biotech Research Institute, Beijing, China) and following the method described by Pickering and Pottinger (Pickering and Pottinger, 1983; Liu et al., 2012). Alkaline phosphatase (AKP) activity was measured according to the procedure described by Bessey et al. (1946) and Dabrowski (1990) using kits from Junshi Biotechnology Co., Ltd., Shanghai, China.
Liver superoxide dismutase (SOD) activities, malondialdehyde (MDA) and total antioxidative capacity (T-AOC) activities
Liver samples were homogenized in ice-cold phosphate buffer (1:10 dilution) (phosphate buffer: 0.064 mol/L, pH 6.4) (Liu et al., 2012). The homogenate was then centrifuged for 20 min (4°C, 3,000×g). An aliquot of the supernatant was used to determine the hepatic SOD, MDA and T-AOC. Hepatic SOD activity and MDA content were measured by the xanthine oxides method (Marklund and Marklund, 1974) and barbituric acid reaction chronometry (Xiang and Wang, 1990), respectively, using kits from Jiancheng Bioengineering Institute, Nanjing, China. Hepatic T-AOC was also evaluated using commercial kits (Jiancheng Bioengineering Institute, Nanjing, China) according to the manufacturer's instructions.
Histology
The fish liver slices were immediately fixed in neutral buffered formalin, embedded in paraffin wax, cut at 5 μm, and stained with haematoxylin and eosin (H & E) as described by Roberts (1989).
Electron microscopy
At the end of the experiment, livers from 3 fish per group were removed for histological examination. Livers were diced into small pieces at approximately 1 mm3, and prefixed in 2.5% glutaraldehyde solution, then fixed in glutaraldehyde overnight at 4°C. After washing in 0.2 M phosphate buffer (pH 7.2) for three 15 min, the samples were post-fixated in cold 1% aqueous osmium tetroxide for 1 h. After rinsing with phosphate buffer again, the specimens were dehydrated in a graded ethanol series of 30–100% and further embedded in Epon 812. The semi-thin sections were stained with 1% toludine blue in 1% borax solution and viewed under a light microscope for initial evaluation. Blocks with specific areas were then chosen and sectioned with glass knives on a KBV ultramicrotome. Eventually, the fifty-nanometer sections of liver were stained with uranyl acetate and lead citrate and observed with H-7650 Transmission Electron Microscope (Hitachi High-Technologies Co., Japan). Ten areas per liver were selected from fish fed the different dietary carbohydrate levels.
Real-time PCR measurement of hepatic HSP70
We used M. amblycephala cDNA sequences in GenBank to design the primers for HSP70 (accession No EU884290.2) and β-actin (accession No AY170122.2) (Ming et al., 2010). The primers were: (1) 5′-CTTTATCAGGGAGGGATGCCAGC-3′ and 5′-CCCTG CAGCATTGAGTTCATAAGGT-3′ for HSP70 cDNA and; (2) 5′-TCGTCCACCGCAAATGCTTCTA-3′ and 5′-CCGTCACCTTCACCGTTCCAGT-3′ for β-actin cDNA. All primers were synthesized by Shanghai Generay Biotech co., LTD. China. The PCR products were 100 to 150 bp long.
We extracted total RNA from the liver tissue using RNAiso Plus (Dalian Takara Co. Ltd., China). RNA samples were treated with RQ1 RNase-Free DNase (Dalian Takara Co. Limited, China) to avoid genomic DNA amplification. We generated cDNA from 500 ng DNase-treated RNA using ExScript RT-PCR Kit (Dalian Takara Co. Ltd., China). The reverse transcription PCR reaction solution consisted of 500 ng RNA, 2 μL 5× Buffer, 0.5 μL dNTP Mixture (10 mM each), 0.25 μL RNase Inhibitor (40 U/μL), 0.5 μL dT-AP Primer (50 mM), 0.25 mL ExScript RTase (200 U/μL), and DEPC H2O, up to a final volume of 10 μL. The reaction conditions were as follows: 37°C for 15 min, 85°C for 5 s, and 4°C thereafter.
We used real-time PCR to determine mRNA levels with an SYBR Green I fluorescence kit (Ming et al., 2010). Real-time PCR was performed in a Mini Opticon Real-Time Detector (Bio-Rad, USA). The fluorescent quantitative PCR reaction solution consisted of 12.5 μL SYBR premix Ex Taq (2×), 0.5 μL PCR Forward Primer (10 μM), 0.5 μL PCR Reverse Primer (10 μM), 2.0 μL RT reaction mix (cDNA solution), 9.5 μL dH2O. The reaction conditions were as follows: 95°C for 2 min, followed by 44 cycles consisting of 95°C for 10 s, 59°C for 20 s, and 72°C for 20 s. The florescent flux was then recorded and the reaction continued at 72°C for 3 min. We measured the dissolution rate between 65 and 92°C. Each increase of 0.2°C was maintained for 1 s and the fluorescent flux was recorded. We calculated the relative quantification of the target gene transcript (HSP70) with a chosen reference gene transcript (β-actin) using the 2−ΔΔCT method (Livak and Schmittgen, 2001). This mathematical algorithm, which does not require a calibration curve, computes an expression ratio based on real-time PCR efficiency and the crossing point deviation of the sample versus a control. We measured the PCR efficiency by constructing a standard curve using a serial dilution of cDNA; ΔΔCT = (CT, Target-CT, β-actin) time x-(CT, Target-CT, β-actin) time 0.
Hypoxia challenge experiment
At the end of the experiment, fifteen fish from each group (5 fish taken from each tank) underwent hypoxia challenge test. The fish were separately placed into six tanks (60 cm×47 cm×40 cm) without aeration. Cumulative mortality was recorded at d 1, d 2, d 3, and d 4. During the challenge test of hypoxia, water temperature was 26.0±1.5°C, pH 6.9±0.1, dissolved oxygen ranged from 0.64 to 0.74 mg/L. The asphyxiant point of Wuchang bream is about 0.5 mg/L (Ou et al., 2001). Oxygen content of water samples was determined using the Winkler method (Strickland and Parsons, 1968).
Statistical analysis
We used SPSS (version 16.0) software Duncan’s multiple range tests and Independent-Samples t-tests to determine the differences. Diverse lower case letters above histogram bars show significant differences (p<0.05) in different carbohydrate groups at each sampling point in Duncan’s multiple range tests. Significant differences (p<0.05) between values obtained from normal and high carbohydrate groups are marked by asterisks above histogram bars in Independent-Samples t-tests. All the results were expressed as means±standard error (X̅ ±SE).
RESULTS
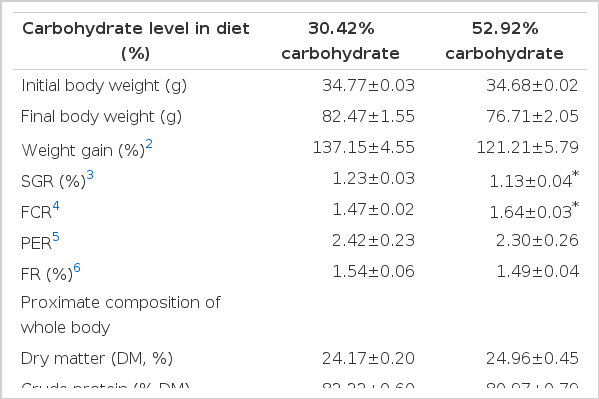
Growth performance and feed utilization of Wuchang bream fed different levels of carbohydrate diet are shown in Table 2. The survival in all treatments was 100%. There was no difference in weight gain between fish fed with the two diets. Protein efficiency ratio (PER) and feeding rate (FR) of Wuchang bream fed 52.92% carbohydrate diet were not statistically significant from those fed with control diet. The fish fed with high carbohydrate diet (52.92%) had significantly lower SGR and higher feed conversion ratio than those of fish fed the control diet (p<0.05). Proximate compositions of whole body for Wuchang bream were also shown in Table 2. There was no difference in dry matter between two dietary treatment groups. Protein, lipid and ash contents in whole body were not significantly affected by the different dietary carbohydrate levels.
Plasma TGL of Wuchang bream fed the control diet for 45 d was significantly lower than that on d 0 (p<0.05). TGL of fish fed 52.92% carbohydrate diet for 90 d was significantly higher than that for 45 d (p<0.05) and also significantly higher than that of fish fed the control diet for 90 d (p<0.05) (Figure 1a). Glucose concentration of fish fed 52.92% carbohydrate diet for 90d was significantly higher than that on d 0 (p<0.05) (Figure 1b). Plasma CHOL of fish fed the control diet for 90 d was significantly higher than those for 0 d and 45 d (p<0.05). There was no difference in CHOL of Wuchang bream between two groups (Figure 1c). Plasma insulin of fish fed the control diet for 45 d was significantly higher than those for 0 d and 90 d, respectively (p<0.05) (Figure 1 d). Plasma cortisol of Wuchang bream fed the control diet for 45 d and 90 d were significantly lower than that for d 0, respectively (p<0.05). Plasma cortisol of Wuchang bream fed high carbohydrate diet for 45 d was significantly lower than that for d 0 (p<0.05). Plasma cortisol of Wuchang bream fed high carbohydrate diet for 90 d was significantly higher than that of fish fed the control diet for 90 d (p<0.05) (Figure 1e). AKP of fish fed the control diet for 90 d was significantly lower than that for 45 d, 0 d, respectively. AKP of Wuchang bream fed high carbohydrate diet was not found significantly different among the 0 d, 45 d, and 90 d samples. AKP of Wuchang bream fed high carbohydrate diet for 45 d was remarkably lower than that of fish fed the control diet for 45 d (p<0.05) (Figure 1f).
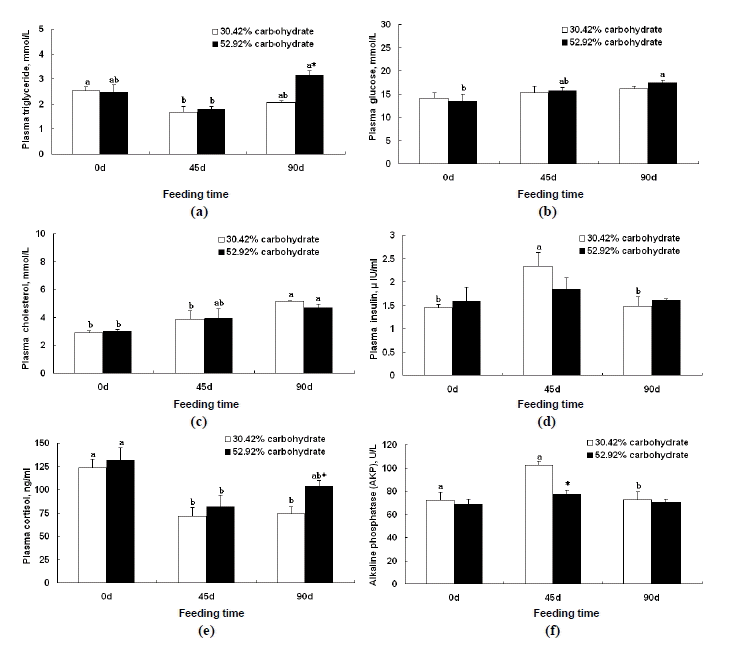
Effects of high or normal carbohydrate diet on plasma triglyceride (a), glucose (b), cholesterol (c), insulin (d), cortisol (e) and alkaline phosphatase (AKP) (f) levels of Wuchang bream (Megalobrama amblycephala). Data are expressed as means±SE (n = 9). Significant differences (p<0.05) between values obtained in 30.42% carbohydrate group and 52.92% carbohydrate group was marked by asterisk by t-tests. Different letters indicate significant differences (p<0.05) in different sampling points by Duncan’s multiple range tests.
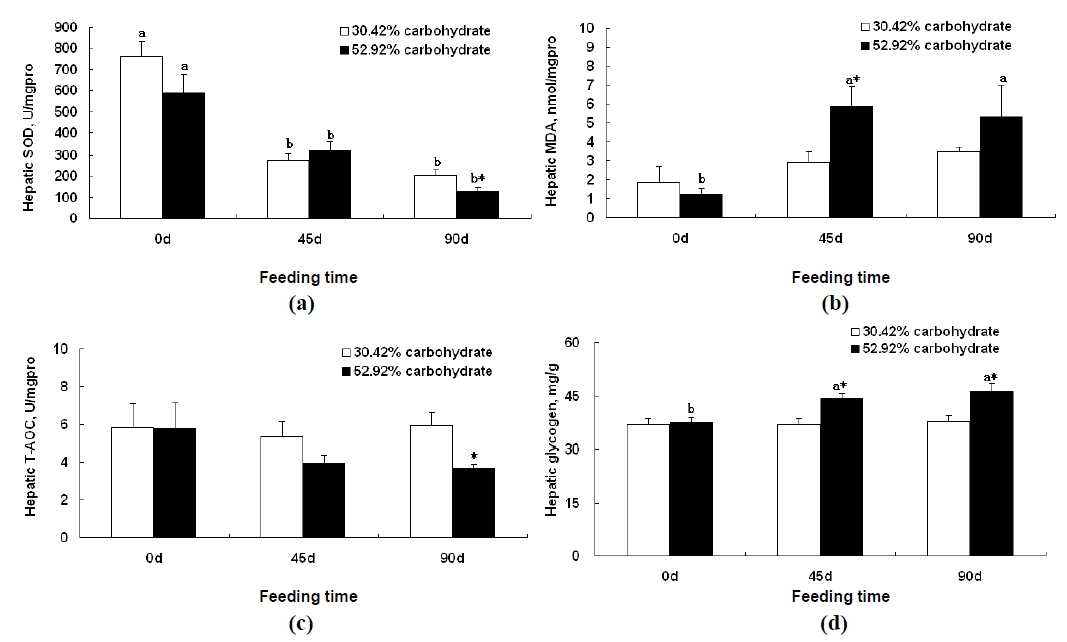
The hepatic SOD activity of Wuchang bream fed the control diet for 45 d and 90 d were significantly lower than that on d 0, respectively (p<0.05). Hepatic SOD activity of Wuchang bream fed high carbohydrate diet for 45 d and 90 d were also significantly lower than that for d0, respectively (p<0.05). Hepatic SOD activity of Wuchang bream fed 52.92% carbohydrate diet for 90 d was significantly lower compared to that of fish fed the control diet for 90 d (p<0.05) (Figure 2a). Hepatic MDA levels of Wuchang bream fed 52.92% carbohydrate diet for 45 d and 90 d were significantly higher than that on d 0, respectively (p<0.05). Hepatic MDA level of Wuchang bream fed 52.92% carbohydrate diet for 45 d was significantly higher compared to that of fish fed the control diet for 45 d (p<0.05) (Figure 2b). The hepatic T-AOC of Wuchang bream fed 52.92% carbohydrate diet for 90 d was significantly lower than that of fish fed the control diet for 90 d (p<0.05) (Figure 2c). The hepatic glycogen content of Wuchang bream fed 52.92% carbohydrate diet for 45 d and 90 d were significantly higher than that for d 0, respectively (p<0.05). Hepatic glycogen content of Wuchang bream fed high carbohydrate diet for 45 d was significantly higher compared to that of fish fed the control diet for 45 d (p<0.05) (Figure 2d). Hepatic glycogen content of Wuchang bream fed 52.92% carbohydrate diet for 90 d was also significantly higher compared to that of fish fed the control diet for 90 d (p<0.05) (Figure 2d).
Liver from fish fed normal carbohydrate diet showed regular-shaped hepatocytes with large centrally located nuclei and some lipidic vacuoles in the cytoplasm which did not disturb the hepatocytes size (Figure 3a). In contrast, fish fed high carbohydrate diet showed foci of non-stained swelling hepatocytes, with numerous varying-size lipid vacuoles. This morphology was more evident for those Wuchang bream fed diets supplemented with the highest dietary inclusion of carbohydrate (Figure 3b).
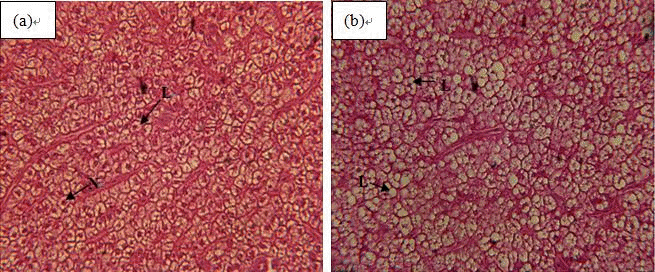
Hepatocytes from Wuchang bream (Megalobrama amblycephala) fed experimental diets. (a) Diet: 30.42% dietary carbohydrate content. Large and spherical nucleus centrally located (H&E, ×400). (b) Diet: 52.92% dietary carbohydrate content. Note swelling hepatocytes with enlarged irregular nucleus located at the periphery of the cell (H&E, ×400). N = Nucleus, L = Lipid droplets.
Hepatocytes of fish fed the control diet showed a great number of small lipid droplets and large nuclei (Figure 4a). On the contrary, fish fed 52.92% carbohydrate diet showed large lipid droplets in a small number that coalesced among them, causing displacement of cellular organelles to the periphery of hepatocytes (Figure 4b).
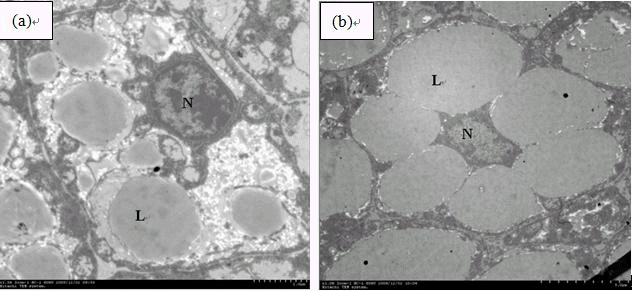
Electron micrographs of Wuchang bream (Megalobrama amblycephala) hepatocytes fed with (a) 30.42% carbohydrate or (b) 52.92% carbohydrate. Note more large lipid droplets in hepatocytes from Wuchang bream (Megalobrama amblycephala) fed with high carbohydrate diet (b). N = Nucleus, L = Lipid droplets.
The relative level of hepatic HSP70 mRNA of Wuchang bream fed 52.92% carbohydrate diet for 90 d was significantly higher than that of fish fed the control diet for 90 d (p<0.05) (Figure 5).
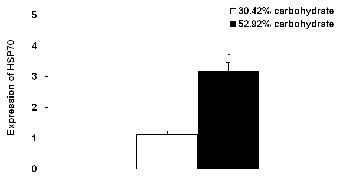
Effects of dietary carbohydrate levels on the relative level of hepatic Heat Shock Protein 70 (HSP70) mRNA of Wuchang bream (Megalobrama amblycephala). Data are expressed as means±SE (n = 9). Significant differences (p<0.05) between values obtained in 30.42% carbohydrate group and 52.92% carbohydrate group was marked by asterisk by t-tests.
There were no significant differences among the cumulative mortality in two groups at 1 d after the hypoxia challenge experiment. But after 2-d hypoxia challenge, the cumulative mortality of the fish in the high carbohydrate group was about 80% while the cumulative mortality in normal groups were about 6.7% (p<0.05). Similarly, after 3-d hypoxia experiment, the cumulative mortality of the fish in the high carbohydrate group was about 100% while the cumulative mortality in normal group was about 86.67%. These indicated that the hypoxia resistance of fish fed the control diet was higher than that of fish fed high carbohydrate diet (Figure 6).
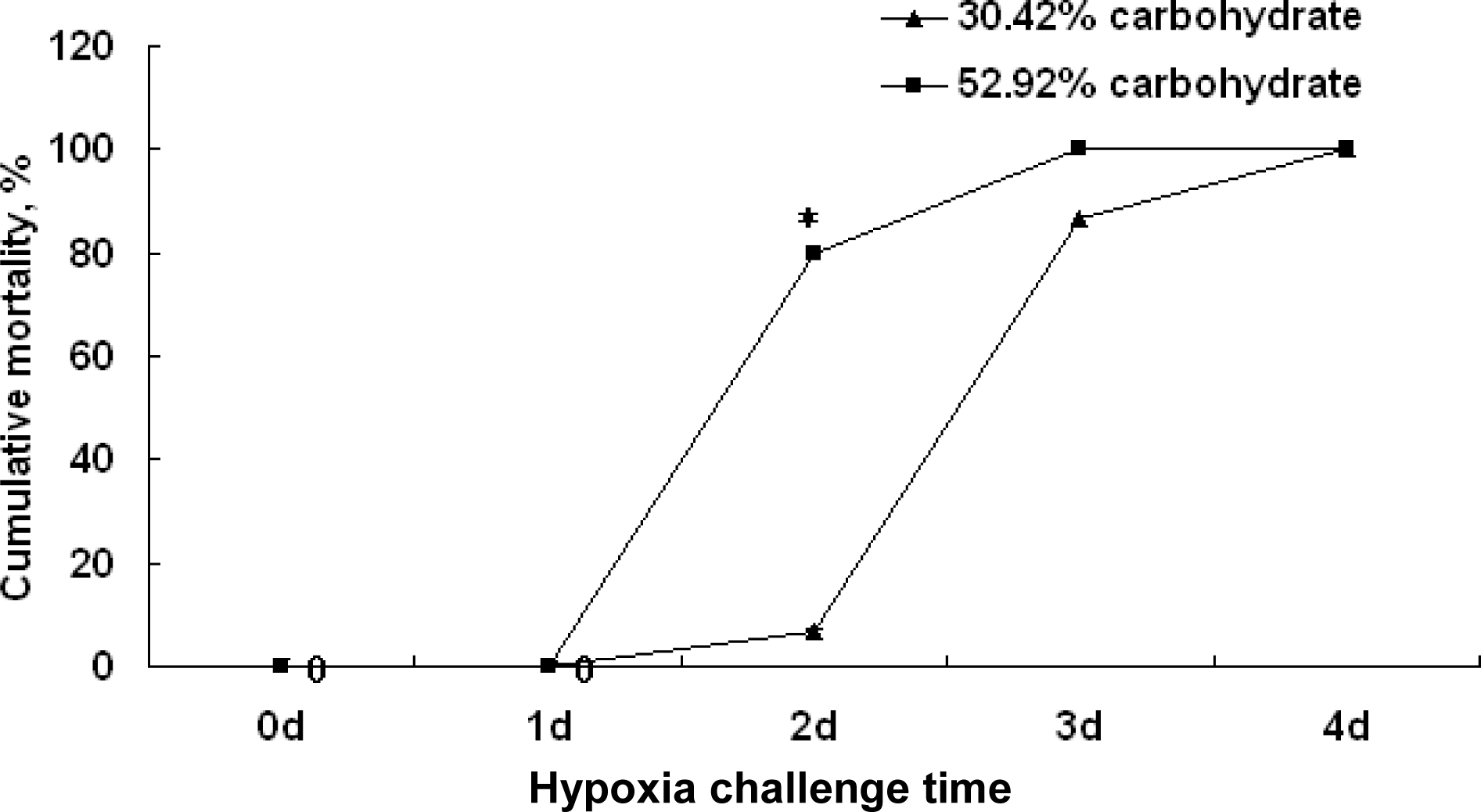
Cumulative mortality after hypoxia challenge in Wuchang bream (Megalobrama amblycephala) fed with the high carbohydrate diet (52.92% carbohydrate) or normal carbohydrate diet (30.42% carbohydrate) for 90 d. Significant differences (p<0.05) between the control and high dietary carbohydrate group at each sampling point after hypoxia challenge are marked by asterisks in t-tests.
DISCUSSION
Previous studies suggested that high dietary carbohydrate levels reduce growth rate and feed utilization (Hemre et al., 2002). In the present study, fish fed high carbohydrate diet had a lower specific growth rate and a higher feed conversion ratio. Similar results were reported previously in herbivorous fish such as grass carp (Ctenopharyngodon idellus) (Gao et al., 2010), omnivorous fish such as gibelio crucian carp (Carassius auratus gibelio) (Tan et al., 2009), as well as in carnivorous fish such as top mouth culter (Erythroculter ilishaeformis Bleeker) (Ge et al., 2007; Liu et al., 2008), southern catfish (Silurua meridionalis) (Luo and Xie, 2010). However, a few studies reported that excessive carbohydrate does not affect growth of rainbow trout (Oncorhynchus mykiss) (Bergot, 1979; Brauge et al., 1994) and Indian major carp (Cirrhinus mrigala) fry (Singh et al., 2006).
In this study, compared with the control group, fish fed high carbohydrate diet for 90 d had significantly higher plasma TGL. Similar findings were obtained in top mouth culter (Erythroculter ilishaeformis Bleeker) (Ge et al., 2007; Liu et al., 2008), European whitefish (Coregonus lavaretus) (Vielma et al., 2003), Atlantic salmon (Salmo salar L.) (Hemre et al., 1995). However, some other studies reported no significant difference by dietary carbohydrate in gilthead sea bream (Sparus aurata) (Enes et al., 2008), largemouth bass (Micropterus salmoides) (Amoah et al., 2008), Atlantic salmon (Salmo salar L.) (Arnesen et al., 1995), and grass carp (Ctenopharyngodon idellus) (Gao et al., 2010).
In addition to changes in growth and intermediate metabolism, excess dietary carbohydrates influence the stress responses of fish (Fletcher, 1997). Stress results in the adrenergic and hypothalamus - pituitary - interrenal (HPI) axis responses, with the HPI response culminating in an increase in plasma cortisol. Therefore the level of cortisol is commonly used as an indicator to assess the stress responses of fish (Barton and Iwama, 1991; Iwama et al., 1997; Wendelaar Bonga, 1997). An increase in dietary carbohydrate from 5% to 30% slightly increased the cortisol levels in Atlantic salmon (Waagbø et al., 1994), but had no apparent effect on rainbow trout (Hilton et al., 1987) or cod (Hemre et al., 1991). In this study, we found that plasma concentrations of cortisol of Wuchang bream fed high dietary carbohydrate was significantly higher than that of fish fed the control diet at the end of the experiment, suggesting that high dietary carbohydrate affected the stress response of Wuchang bream.
The stress response might also increase free radical contents, leading to increased lipid peroxidation and lipid peroxidation injury (Zhou and Liang, 2003; Chen et al., 2006; He et al., 2007). In Atlantic salmon, dietary intake of high carbohydrate can impact on the immunity and fish mortality after Aeromonas Salmonicida infection (Waagbø et al., 1994). The hepatic lipid peroxide will decompose further to produce a large amount of aldehydes, alcohols and hydrocarbon of which MDA is a component with strong toxicity. SOD plays a significant role in resisting oxide damage (Gu et al., 1995). In this study, compared with control group, fish fed a high carbohydrate diet had significantly lower hepatic SOD activity for 90 d, higher hepatic MDA activities for 45d, and higher hepatic T-AOC for 90 d. These results suggest that excessive carbohydrate lowered the hepatic antioxidant abilities of Wuchang bream.
The fate of excess hepatic carbohydrate is either towards glycogen synthesis or towards lipogenesis (Enes et al., 2006a). In this study, we observed that hepatic glycogen content of Wuchang bream fed a high carbohydrate diet was significantly higher compared to that of fish fed the control diet, similar to a previous study (Yengkokpam, 2003). In the present study, high carbohydrate diet affected hepatocyte morphology. We observed the hepatocytes of fish fed a high carbohydrate diet showed large lipid droplets that coalesced among them, causing displacement of cellular organelles to the periphery of hepatocytes. This observation agrees with reports of hepatocyte hypertrophy due to increased level of carbohydrate in the feed of L. rohita juveniles (Kumar et al., 2005) and L. rohita fry (Mohapatra et al., 2003).
Heat shock proteins (HSPs) are one of the most conserved and important protein families, present in all organisms including fish (Basu et al., 2002). HSP70 is mainly involved in stress protection, improving cell survival and raising tolerance to environmental stressors or harm (Basu et al., 2002). Thus, HSP70 has been most widely chosen as a bioindicator of stress. In this study, the relative level of hepatic HSP70 mRNA of Wuchang bream fed high carbohydrate diet for 90 d was significantly higher than that of fish fed the control diet for 90 d. The finding of our study indicated that high dietary carbohydrate may lead to metabolic stress in M. amblycephala, similar to a previous study (Enes et al., 2006b).
The objective of hypoxia challenge was to determine the effects of hypoxia on the mortality of the fish fed high dietary carbohydrate. Inadequate dissolved oxygen in the aquatic environment is a well-established cause of fish mortality (Choi et al., 2007). Similar experiments were carried out on estuarine fish (Leiostomus xanthurus) (Cooper et al., 2002) and tilapia (Oreochromis spp.) (Bunch and Bejerano, 1997; Evans et al., 2003). Fish fed with high carbohydrate diet are assumed to be under constant metabolic stress (Pieper and Pfeffer, 1980; Fletcher, 1981), which may lead to suppressed immune functions (Ellis, 1981; Maule et al., 1989; Wiik et al., 1989; Vielma et al., 2003; Kumar et al., 2005). In this study, after hypoxia challenge, the mortality was higher in the high dietary carbohydrate group compared to the control group, at d 2 or d 3 of challenge. It was considered that the disease resistance of the fish fed high dietary carbohydrate was lower than those of the fish fed control diet. Excessive dietary carbohydrate might have lowered the resistance to hypoxia.
CONCLUSIONS
In summary, based on results from growth performances, blood biochemical parameters, hepatic antioxidation enzyme activities, the change of hepatic morphology, the relative level of hepatic HSP70 mRNA, and the hypoxia challenge, high dietary carbohydrate reduced the growth rate and feed utilization, decreased fish resistance to hypoxia challenge, therefore health status. These findings provide a better understanding of the health implications of high dietary carbohydrate for Wuchang bream.
Acknowledgements
We thank Dr. Ya-Xiong Tao at Auburn University in United States for advice and editing during preparation of the manuscript. This work was supported by Modern Agro-industry Technology Research System, China (CARS-46), Special Fund for Agro-scientific Research in the Public Interest of China (201003020), Open Fund of Key Laboratory of Freshwater Fisheries and Germplasm Resources Utilization of the Ministry of Agriculture (KF201311), Freshwater Fisheries Research Center, Chinese Academy of Fishery Sciences, China Central Governmental Research Institutional Basic Special Research Project from Public Welfare Fund (2009JBFB16).