2. Zhao X, Xing T, Chen X, et al. Yield, thermal denaturation, and microstructure of proteins isolated from pale, soft, exudative chicken breast meat by using isoelectric solubilization/precipitation. Process Biochem 2017; 58:167–73.

3. Petracci M, Bianchi M, Mudalal S, et al. Functional ingredients for poultry meat products. Trends Food Sci Technol 2013; 33:27–39.

4. Chen H, Wang HH, Qi J, et al. Chicken breast quality–normal, pale, soft and exudative (PSE) and woody–influences the functional properties of meat batters. Int J Food Sci Technol 2018; 53:654–64.

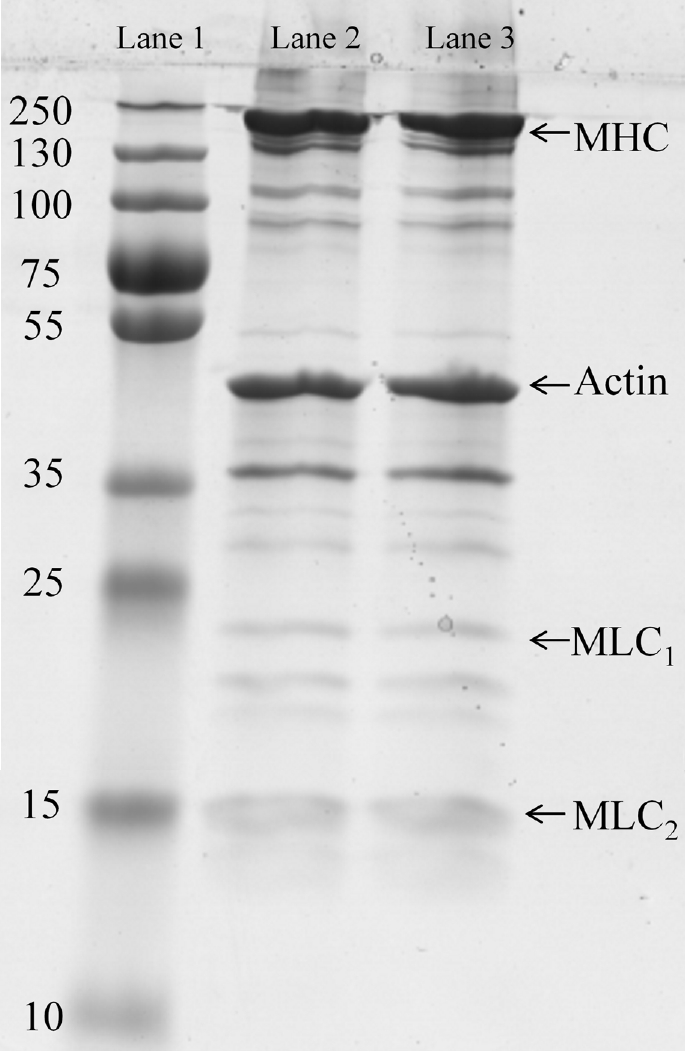
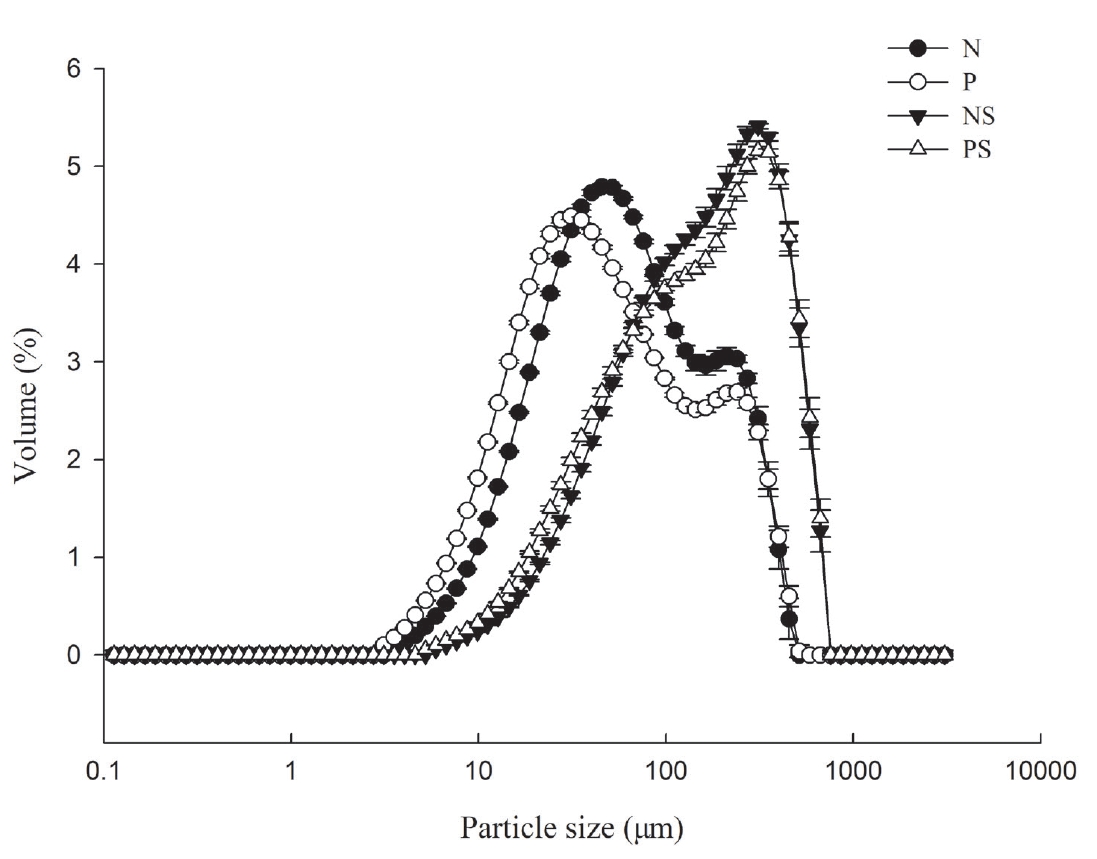
5. Li K, Chen L, Zhao YY, et al. A comparative study of chemical composition, color, and thermal gelling properties of normal and PSE-like chicken breast meat. CyTA-J Food 2015; 13:213–9.

7. Sun XD, Holley RA. Factors influencing gel formation by myofibrillar proteins in muscle foods. Compr Rev Food Sci Food Saf 2011; 10:33–51.

8. Wang H, Pato MD, Shand PJ. Biochemical properties of natural actomyosin extracted from normal and pale, soft, and exudative pork loin after frozen storage. J Food Sci 2005; 70:C313–20.

9. Liu R, Zhao S, Yang H, et al. Comparative study on the stability of fish actomyosin and pork actomyosin. Meat Sci 2011; 88:234–40.


10. Camou JP, Sebranek JG. Gelation characteristics of muscle proteins from pale, soft, exudative (PSE) pork. Meat Sci 1991; 30:207–20.


11. Wang H, Pato M, Pietrasik Z, Shand P. Biochemical and physicochemical properties of thermally treated natural actomyosin extracted from normal and PSE pork
Longissimus muscle. Food Chem 2009; 113:21–7.

13. Liu R, Zhao SM, Xie BJ, et al. Contribution of protein conformation and intermolecular bonds to fish and pork gelation properties. Food Hydrocoll 2011; 25:898–906.

14. Zhao X, Xing T, Chen X, et al. Changes of molecular forces during thermo-gelling of protein isolated from PSE-like chicken breast by various isoelectric solubilization/precipitation extraction strategies. Food Bioproc Tech 2017; 10:1240–7.

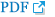
15. Ni N, Wang Z, He F, et al. Gel properties and molecular forces of lamb myofibrillar protein during heat induction at different pH values. Process Biochem 2014; 49:631–6.

16. Ogawa M, Nakamura S, Horimoto Y, et al. Raman spectroscopic study of changes in fish actomyosin during setting. J Agric Food Chem 1999; 47:3309–18.


17. Gornall AG, Bardawill CJ, David MM. Determination of serum proteins by means of the biuret reaction. J Biol Chem 1949; 177:751–66.


19. Li K, Kang ZL, Zhao YY, et al. Use of high-intensity ultrasound to improve functional properties of batter suspensions prepared from PSE-like chicken breast meat. Food Bioproc Tech 2014; 7:3466–77.

20. Chen H, Han M. Raman spectroscopic study of the effects of microbial transglutaminase on heat-induced gelation of pork myofibrillar proteins and its relationship with textural characteristics. Food Res Int 2011; 44:1514–20.

21. Kocher P, Foegeding E. Microcentrifuge-based method for measuring water holding of protein gels. J Food Sci 1993; 58:1040–6.

22. Ellman GL. Tissue sulfhydryl groups. Arch Biochem Biophys 1959; 82:70–7.


25. Eadmusik S, Molette C, Fernandez X, et al. Are one early muscle pH and one early temperature measurement sufficient to detect PSE breast meat in turkeys? Br Poult Sci 2011; 52:177–88.


26. Pighin DG, Sancho AM, Gonzalez CB. Effect of salt addition on the thermal behavior of proteins of bovine meat from Argentina. Meat Sci 2008; 79:549–56.


27. Feng M, Pan L, Yang X, et al. Thermal gelling properties and mechanism of porcine myofibrillar protein containing flaxseed gum at different NaCl concentrations. LWT-Food Sci Technol 2018; 87:361–7.

28. Liu J, Schwartzkopf M, Arner A. Rigor bonds cause reduced sarcomeric volume in skinned porcine skeletal muscle under PSE-like conditions. Meat Sci 2018; 139:91–6.


29. Lesiów T, Xiong YL. A simple, reliable and reproductive method to obtain experimental pale, soft and exudative (PSE) pork. Meat Sci 2013; 93:489–94.


30. Liu MN, Foegeding EA, Wang SF, et al. Denaturation and aggregation of chicken myosin isoforms. J Agric Food Chem 1996; 44:1435–40.

31. Park JW, Lanier TC. Scanning calorimetric behavior of tilapia myosin and actin due to processing of muscle and protein purification. J Food Sci 1989; 54:49–51.

32. Ishioroshi M, Jima KS, Yasui T. Heat-induced gelation of myosin: factors of pH and salt concentrations. J Food Sci 1979; 44:1280–4.

33. Wright DJ, Wilding P. Differential scanning calorimetric study of muscle and its proteins: myosin and its subfragments. J Sci Food Agric 1984; 35:357–72.


34. Wilhelm AE, Maganhini MB, Hernández-Blazquez FJ, et al. Protease activity and the ultrastructure of broiler chicken PSE (pale, soft, exudative) meat. Food Chem 2010; 119:1201–4.

35. D’Alessandro A, Zolla L. Meat science: From proteomics to integrated omics towards system biology. J Proteomics 2013; 78:558–77.


36. Chen L, Li C, Ullah N, et al. Different physicochemical, structural and digestibility characteristics of myofibrillar protein from PSE and normal pork before and after oxidation. Meat Sci 2016; 121:228–37.


37. Xiong YL, Bacnchard SP. Myofibrillar protein gelation: viscoelastic changes related to heating procedures. J Food Sci 1994; 59:734–8.

38. Egelandsdal B, Fretheim K, Samejima K. Dynamic rheological measurements on heat-induced myosin gels: effect of ionic strength, protein concentration and addition of adenosine triphosphate or pyrophosphate. J Sci Food Agric 1986; 37:915–26.

39. Zhao YY, Wang P, Zou YF, et al. Effect of pre-emulsification of plant lipid treated by pulsed ultrasound on the functional properties of chicken breast myofibrillar protein composite gel. Food Res Int 2014; 58:98–104.

40. Xu Y, Xia W, Jiang Q. Aggregation and structural changes of silver carp actomyosin as affected by mild acidification with d-gluconic acid δ-lactone. Food Chem 2012; 134:1005–10.


41. Chan JK, Gill TA. Thermal aggregation of mixed fish myosins. J Agric Food Chem 1994; 42:2649–55.

42. Lanier TC, Carvajal P, Yongsawatdigul J. Surimi gelation chemistryPark JW, editorSurimi and surimi seafood. Boca Raton, FL, USA: CRC press; 2013. p. 101–39.
43. Ding Y, Liu R, Rong J, et al. Rheological behavior of heat-induced actomyosin gels from yellowcheek carp and grass carp. Eur Food Res Technol 2012; 235:245–51.

44. Lesiów T, Xiong YL. Mechanism of rheological changes in poultry myofibrillar proteins during gelation. Avian Poult Biol Rev 2001; 12:137–49.

45. Sano T, Ohno T, Otsuka-Fuchino H, et al. Carp natural actomyosin: thermal denaturation mechanism. J Food Sci 1994; 59:1002–8.

46. Sankar TV, Ramachandran A. Thermal stability of myofibrillar protein from Indian major carps. J Sci Food Agric 2005; 85:563–8.

47. Liu R, Zhao S, Regenstein JM, et al. Gelling properties of fish/pork mince mixtures. J Food Sci 2016; 81:C301–7.


48. Xu Y, Xia W, Yang F, et al. Protein molecular interactions involved in the gel network formation of fermented silver carp mince inoculated with
Pediococcus pentosaceus
. Food Chem 2010; 120:717–23.








 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print







